INTRODUCTION
Vertigo is a common presenting symptom in the emergency department (ED). Vertigo has both peripheral and central causes, with most vertigo cases being peripheral (inner ear) in etiologies [1-3]. Common causes of peripheral vertigo include benign paroxysmal positional vertigo (BPPV), vestibular neuritis, labyrinthitis, and Meniere disease. Peripheral vertigo is often considered benign, and patients with this diagnosis are often discharged from the ED after symptomatic treatments.
Dizziness or vertigo is one of the most common categories of symptoms for visiting the ED in the United States [4]. Dizziness or vertigo is very common in the general population, with one in five people of working age and one in four elderly people age having experienced the symptom during the past month [5,6]. There are more than 2 million US ED visits annually for dizziness or vertigo, comprising approximately 4.4% of all chief complaints in awake patients [4,7-9]. The number of these visits increased by 90% between 1995 and 2009, and the national cost of their evaluations in the US ED was 3.9 billion US dollars per year in 2011 compared with 1.1 billion US dollars per year in 1993 [10,11].
Vestibular asymmetry has been associated with an increased risk of falls and fractures. In addition, various prescriptions to control the related symptoms, such as anticholinergics, antiemetics, benzodiazepines and others, might also increase the risk of falls and fractures [12-16]. Therefore, it is possible that ED visits for peripheral vertigo are associated with an increased risk of various types of injury after discharge, and this hypothetical risk may last a long time, as peripheral vertigo tends to frequently recur. However, studies demonstrating this association, especially with long-term perspectives, are lacking. Therefore, we conducted a population-based observational study using a nationally representative cohort. The purpose of the study was to determine whether a visit to an ED for a new episode of peripheral vertigo significantly increased the risk of injury.
METHODS
Study design
We conducted a matched-cohort study using a nationally representative dataset. The main exposure was an ED visit for new peripheral-type vertigo. Incidence density sampling was used to match each patient with an exposure to five unexposed individuals (also without previous injury) [17]. The primary outcome was a new injury event within 1 year after an ED visit. The institutional review boards of the Seoul National University Bundang Hospital approved the study (X-1705/399-904) and provided a waiver of informed consent.
Data source
The data source was the National Health Insurance Service–National Sample Cohort (NHIS-NSC) dataset released in 2015 by the Korean National Health Insurance Service (NHIS) [18]. The dataset was obtained from a population-based cohort with de-identified claim information including 1 million randomly sampled individuals (using stratification), from a real Korean population. The dataset contains diagnostic codes based on the Korean Classification of Diseases 6 (a Korean modification of the International Classification of Diseases-10 coding system), prescription and procedure codes, and related costs, as well as demographic information such as age, sex, and socioeconomic status from 2002 to 2013. The database also has information about disability and death based on the national disability registration data and death certificates.
Definition of exposure and outcome events
The exposure cohort included adult patients (aged 20 years or older) visiting the ED with a principal diagnosis of vertiginous disease of peripheral origin, including Meniere disease (H81.0x), BPPV (H81.1x), vestibular neuronitis (H81.2x), labyrinthitis (H83.0x), and other peripheral vertigo (H81.3x); but no new diagnosis of central vertigo (H81.4x) within the week following the ED visit. The primary outcome was any new incident of injury regardless of the ED visit. The secondary outcomes were specific types of injuries, including injuries to the head and neck (S0.x, S1.x); brain (intracranial [S06.x]); trunk (thorax [S2.x] and abdomen [S3.x]); upper extremity (shoulder and upper arm [S4.x], elbow and forearm [S5.x], and wrist, hand, and fingers [S6.x]); and lower extremity (hip and thigh [S7.x], knee and lower leg [S8.x], and ankle and foot [S9.x]).
Construction of cohorts and included covariates
The study population included in this analysis was based on the following criteria. We first excluded all individuals with a diagnosis of peripheral vertigo or injury from January 1, 2002, to December 31, 2003, before study entry (January 1, 2004). Among the remaining eligible participants, we built our exposure and comparison cohorts using incidence density sampling without replacement [17]. Specifically, we identified the first visit of participants who visited the ED for a vertiginous disorder of peripheral origin without any previous or concurrent injury event; then, the number of days from their study entry (the day of initial diagnosis) to the first event of injury was determined. We then randomly matched each of the exposed patients to five unexposed individuals who also had no previous injury, and were alive in the NHIS cohort for at least the same period based on the age (±1 year), sex, and household income level (±1 level). Resampling was not allowed, and individuals initially selected as an unexposed group could visit the ED for peripheral vertigo at a later time. In both cohorts, observation was censored at the time of their first injury event, death, or the end of the NHIS cohort follow-up (December 31, 2013).
The covariates considered were age, sex, household income level, and comorbidities including diabetes mellitus, hypertension, ischemic heart disease, stroke, heart failure, chronic renal failure, advanced liver disease, chronic obstructive pulmonary disorder, peripheral arterial disease, and malignancy. The presence of each of the comorbidities was defined based on the claim information of the 2-year period preceding the exposure [19-23].
Statistical analyses
Categorical variables were reported using frequencies and proportions. Continuous variables were reported using mean and standard deviation or median and interquartile range, as appropriate. Wilcoxon rank-sum test, the chi-square test, or Fisher exact test was performed as appropriate for comparisons between groups.
We used an extended Cox model using time-varying coefficients to model the subsequent risk of injury. Specifically, we estimated the hazard ratios (HRs) and their 95% confidence intervals (CIs) for injury at three post-visit intervals (first month, 1 to 3 months, and 3 months to 1 year) to capture the diminishing effect size of acute peripheral vertigo episode [24]. Using the method, we assessed the unadjusted and adjusted effect of the exposure. For the adjustment, we used two sets of covariates, one including age and sex and the other including all the covariates including age, sex, household income and whole set of comorbidities.
The goodness-of-fit of the multivariable models was tested with Gronnesby and Borgan goodness-of-fit test. The proportional hazard assumption was assessed by examination of Schoenfeld residual plots. The results of the Cox regression analyses were presented as HRs, with their 95% CIs. P-values <0.05 were considered significant. All data handling and statistical analyses were performed using R-packages ver. 3.3.2 (R Foundation for Statistical Computing, Vienna, Austria).
RESULTS
From 2004 to 2013, a total of 776 patients were included as the exposure cohort, and 3,880 patients were included as the comparison cohort (Fig. 1 and Table 1). The prevalence of hypertension, dyslipidemia, ischemic heart disease, and stroke was significantly higher in the exposure cohort (P<0.001, P=0.001, P<0.001, and P=0.006, respectively) than in the comparison cohort. The median duration of follow-up was 365 (interquartile range, 365– 365) days for both cohorts (P=0.623), with almost the same proportion of full observation over 3 years (n=727, 93.7% vs. n=3,653, 94.1%, respectively; P=0.677).
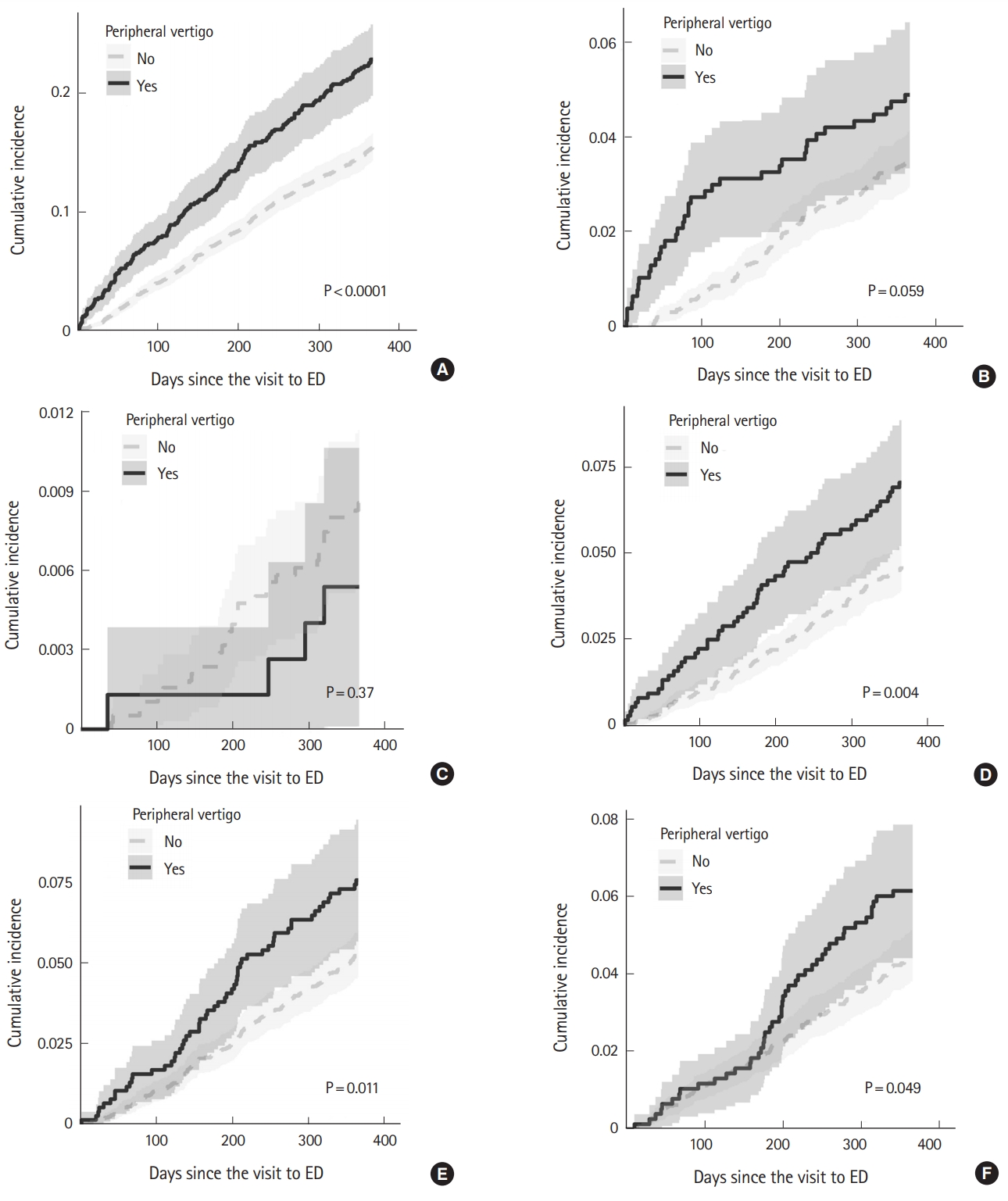
A total of 170 patients (21.9%) in the exposure cohort had any injury at least once during the 1-year follow-up period (Table 2), while 577 (14.9%) had such an event in the reference cohort (P<0.001). With several types of injuries, a statistically significant difference occurred between the groups: trunk injury (53 [6.8%] vs. 173 [4.5%], respectively; P=0.007); and upper extremity injury (57 [7.3%] vs. 199 [5.1%], respectively; P<0.017). Fig. 2 shows the cumulative incidence of the first injury events during the follow-up period.
The associations between peripheral vertigo and injury during the post-visit intervals (first month, 1 to 3 months, and 3 months to 1 year) were assessed using extended Cox regression. We built three regression models: 1) a univariable model; 2) a multivariable model including peripheral vertigo, age group, and sex; and 3) a full multivariable model including peripheral vertigo, age group, sex, household income level, and comorbidities. In all the three models, acute peripheral vertigo was associated with an increased risk of injury. The HRs in the first month were 5.33 (95% CI, 2.89–9.83; P<0.001); 5.33 (95% CI, 2.89–9.83; P<0.001); and 5.23 (95% CI, 2.83–9.64; P<0.001), respectively. The HRs for the 1 month to 3 months were 1.53 (95% CI, 1.04–2.24; P=0.030); 1.53 (95% CI, 1.04–2.24; P=0.030); and 1.50 (95% CI, 1.02–2.20; P=0.039), respectively. For the 3 months to 1 year HRs, these were 1.40 (95% CI, 1.14–1.72; P=0.001); 1.40 (95% CI, 1.14–1.72; P=0.001); and 1.37 (95% CI, 1.11–1.68; P=0.003), respectively (Table 3). Fig. 3 shows both the crude and adjusted cumulative incidence of injury in the exposure and reference cohorts during the observation. The only other variable associated with the injury risk was history of stroke (Supplementary Tables 1, 2) with a HR of 1.53 (95% CI, 1.02–2.29; P=0.039).
DISCUSSION
In this population-based longitudinal study, we found a new ED visit for acute peripheral vertigo to be a significant risk factor for injury for up to a year. We also found that the effect size of the exposure is time-dependent with a decreasing effect. To the best of our knowledge, this is the first observational study reporting the association between ED visits for peripheral vertigo and future risk of injury.
Current evidence on the association between peripheral vertigo and the future risk of injury is lacking. Two administrative data-based studies are currently available. The first one assessed the risk of any kind of fracture in a Taiwanese population of patients with new BPPV compared to age-, sex-, and index year-matched, randomly selected controls for up to 12 years [25]. In the study, the authors observed a 1.14-fold higher risk of fracture after the diagnosis of peripheral vertigo. The second study compared the risk of accidental injuries after ED visit for peripheral vertigo to that of renal colic in a population dataset of Canadian ED visitors [26]. In contrast to the first study, no significant association was observed.
Our study demonstrated a significant increase in the risk of injury for up to a year after an ED visit for peripheral vertigo. This suggests the need for ED physicians to alert patients and their families about the risk, especially if the patient has a higher baseline risk of injury. The other risk factors considered for fall, especially in the elderly population, are frailty, vision, balance or gait problems, postural hypotension, and various medications [27-29]. Recommendations of some preventive measures to prevent falls, such as improvement of the patient’s environment (installation of handles and removal of tripping hazards such as slippery and uneven surfaces), physical training, medication adjustment, or proper use of assistive devices may be helpful if provided by ED physicians for patients with vertigo as well as multiple risk factors for fall [29-33].
Possibly, appropriate help from guardians might be crucial in preventing injury. However, we could not assess its effect as our dataset does not provide such details. Currently, there has been no study assessing whether such external help could significantly reduce the risk of injury. The authors hope to see a future study shedding light on this subject.
This study has several limitations. First, misclassification is a potential cause of bias in a population-based study using claims records. Secondly, the diagnosis of peripheral vertigo made in the EDs is often not final and can often change later after the followup. Thirdly, there was no information available on symptom intensity or duration. Fourth, we did not assess the effect of medication on the outcomes because we thought the sample size of the study population was not large enough to analyze the effect of individual pharmacological variation.
Despite these limitations, the study also has some strengths. This is the first population-based analysis demonstrating that a new ED visit for peripheral vertigo is a risk factor for future injury. We analyzed data over a sufficiently long period using a statistical method that can model the time-dependency of effect size. Finally, we assessed various types of injury risks, including brain injury, which were not analyzed in previous studies.
In conclusion, visiting the ED for acute peripheral vertigo was associated with an increased risk of injury for up to a year. It would be prudent to alert the patients and their family members about the risk, and recommend relevant preventive measures.