INTRODUCTION
Reexpansion pulmonary edema (RPE) is a complication of pleural drainage that is frequently encountered after decompression of pneumothorax or pleural effusion. It is a potentially lethal condition, with a reported mortality rate of 20% [1]. Therefore, physicians should consider the possibility of RPE following pleural drainage. Most reported cases involve ipsilateral RPE [2-4]; however, the pathophysiologic mechanism of contralateral RPE remains unclear. We report a case of contralateral RPE after pleural drainage and compare its progression to that of previously reported cases to better understand the pathophysiology of this complication.
CASE REPORT
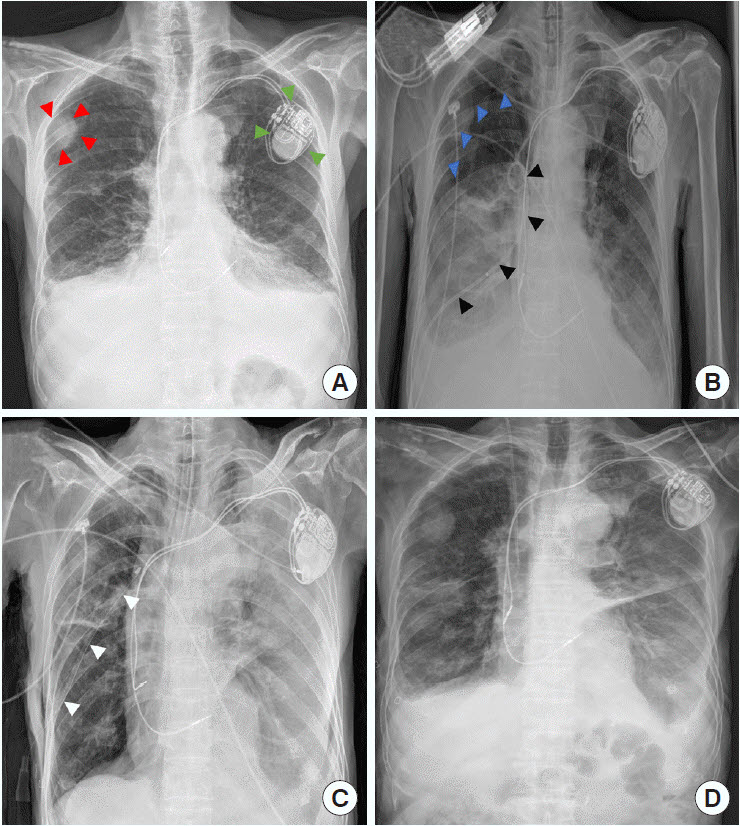
An 83-year-old male patient who underwent pacemaker implantation for sinus node dysfunction nearly 4 years ago presented to the emergency department with a complaint of dyspnea for 3 days. At presentation, his blood pressure, heart rate, respiratory rate, body temperature, and oxygen saturation were 68 mmHg, 92 beats/min, 30 breaths/min, 36.2°C, and 93%, respectively. Blood gas analysis showed arterial oxygen tension of 70 mmHg, arterial carbon dioxide tension of 29 mmHg, and blood pH of 7.46. Complete blood count (CBC) analysis revealed a white blood cell (WBC) count, hemoglobin (Hb) level, and platelet count of 7,790/µL, 12.5 g/dL, and 268,000/µL, respectively. Costophrenic angle blunting was observed in both hemithoraces on chest radiograph (CXR) (Fig. 1A). We confirmed the presence of a large pleural effusion in the right hemithorax using sonography, whereas there was minimal pleural effusion in the left hemithorax.
Oxygen administered to the patient via a nasal prong helped stabilize the vital signs and relieve dyspnea. Thereafter, diagnostic thoracentesis was performed and revealed lymphocyte predominance and high adenosine deaminase level. A clinical diagnosis of tuberculous pleurisy was established, and antitubercular medication was initiated. Despite good therapeutic compliance, the pleural effusion was not improved. Therefore, percutaneous drainage with a pigtail catheter (8.5 Fr) was performed on hospitalization day 5, and 150 mL of fluid was drained. After approximately 15 minutes, the patient’s condition deteriorated, and his vital signs decreased further. Despite a high-flow nasal cannula, the patient’s respiratory rate was 60 breaths/min; therefore, endotracheal intubation was performed at 45 minutes after percutaneous drainage insertion. A follow-up CXR showed partial collapse of the affected lung with pneumothorax (Fig. 1B).
Although the chest tube drained 1,300 mL of fluid, the patient’s oxygen demand remained high. A repeat CXR was performed and revealed partial expansion of the affected lung and severe infiltrative patterns in the unaffected lung, suggesting contralateral RPE (Fig. 1C). Blood gas analysis showed arterial oxygen tension of 89 mmHg, arterial carbon dioxide tension of 52 mmHg, and blood pH of 7.42 while on a ventilator with 60% oxygen delivery and 10 mmHg of positive end-expiratory pressure. Follow-up CBC analysis showed WBC count of 14,500/µL, Hb level of 11.4 g/dL, and platelet count of 258,000/µL. To avoid exacerbation, the patient was transferred to the intensive care unit. The patient’s oxygenation and fraction of inspired oxygen requirement improved over the next 48 hours. Repeat CBC analysis revealed WBC count of 9,900/µL, Hb level of 11.1 g/dL, and platelet count of 156,000/µL. The patient was successfully extubated, and his CXR revealed an improvement in RPE 2 days after pigtail catheterization (Fig. 1D).
The study was conducted according to the guidelines of the Declaration of Helsinki and approved by the Institutional Review Board of Chungnam National University Hospital (No. 2021-07-098). The need for informed consent was waived as the extracted data included clinical data that did not have any personally identifiable information.
DISCUSSION
Although several studies have reported cases of RPE, its etiopathogenesis is still not fully understood. RPE development might be attributed to a combination of inflammatory responses initiated by lung reinflation and altered hydrostatic forces [1]. This promotes interstitial fluid shift, impairs ventilation, and leads to a clinical syndrome that presents as sudden, dramatic hypoxia that is unresponsive to oxygen supplementation [5].
Her and Mandy [6] suggested that contralateral RPE can be caused by a systemic microvascular injury through a leukocyte-mediated inflammatory reaction. However, the cases considered in their report showed subacute contralateral RPE between 24 and 48 hours after primary drainage of an ipsilateral hemithorax, contrary to the acute onset (within few hours) of contralateral RPE in our case. In addition, leukopenia, which was present in previous cases, was not observed for more than 48 hours after the acute lung injury in our case. Therefore, the hypothesis that contralateral RPE is a leukocyte-mediated acute lung injury was not supported by our findings. Conversely, another case report by Kim et al. [7] reported that contralateral RPE acutely occurred after massive drainage of 1,500 mL; their findings also did not support the theory of leukocyte-mediated contralateral RPE suggested by Her and Mandy [6].
Our case shares several clinical findings with those of Kim et al. [7]’s case. First, acute contralateral RPE occurred within a few hours after primary drainage of the affected lung. Second, the affected lung was not fully expanded due to co-existing pneumothorax and remaining pleural effusion when acute contralateral RPE occurred. Physiologically, net fluid transfer across the pulmonary capillaries depends on the difference between the hydrostatic and colloidal osmotic pressures in the normal physiological state and the permeability of the capillary membrane (Starling’s law) [8]. However, some pathophysiologic states can cause redirection of blood flow to an uninvolved area, increasing the hydrostatic pressure unilaterally and resulting in contralateral pulmonary edema [9]. A previous report stated that an ipsilateral increase in hydrostatic pressure due to jet flow of mitral valve regurgitation can lead to unilateral pulmonary edema [10]; thus, we suggest that contralateral RPE acutely occurring after massive pleural drainage can be caused by acutely increased unilateral hydrostatic pressure rather than by an inflammatory reaction, such as leukocyte-mediated lung injury.
In summary, considering our serial CBC findings and the pathophysiology of unilateral pulmonary edema by ipsilaterally increased hydrostatic pressure, we suggest that the dissymmetric hydrostatic pressure scenario is more suitable in the acute onset of contralateral RPE after massive pulmonary drainage. Further research is required to confirm the pathophysiology of RPE, which can help physicians avoid this emergency in clinical practice.