Intra-arrest transesophageal echocardiography during cardiopulmonary resuscitation
Article information
Abstract
Determining the cause of cardiac arrest (CA) and the heart status during CA is crucial for its treatment. Transesophageal echocardiography (TEE) is an imaging method that facilitates close observation of the heart without interfering with cardiopulmonary resuscitation (CPR). Intra-arrest TEE is a point-of-care ultrasound technique that is used during CPR. Intra-arrest TEE is performed to diagnose the cause of CA, determine the presence of cardiac contraction, evaluate the quality of CPR, assist with catheter insertion, and explore the mechanism of blood flow during CPR. The common causes of CA diagnosed using intra-arrest TEE include cardiac tamponade, aortic dissection, pulmonary embolism, and intracardiac thrombus, which can be observed on a few simple image planes at the mid-esophageal and upper esophageal positions. To operate an intra-arrest TEE program, it is necessary to secure a physician who is capable of performing TEE, provide appropriate training, establish implementation protocols, and prepare a plan in collaboration with the CPR team.
INTRODUCTION
Transesophageal echocardiography (TEE) is a diagnostic tool that can obtain images of the heart from its nearest location. It provides numerous types of echocardiographic information, including two-dimensional or three-dimensional images, M-mode, color flow imaging, Doppler studies, and related calculations, such as transthoracic echocardiography (TTE). Compared with TTE, TEE provides excellent echocardiographic windows to the heart regardless of the patient’s body type. Despite its advantages, TEE has rarely been used in patients with cardiovascular emergencies in the emergency department (ED) when it was initially introduced into clinical practice. TEE in the ED has been used in urgent situations such as cardiac arrest (CA). Recent cardiopulmonary resuscitation (CPR) guidelines recommend or suggest the use of point-of-care ultrasound as a method to determine the reversible cause of CA during CPR, thus encouraging the use of echocardiography during the performance of advanced life support [1,2].
TTE is an easy and simple method for point-of-care ultrasound during resuscitation. However, it is not suitable for close inspection of the heart while chest compressions are being performed. By employing TEE, rescuers can continuously observe the heart without interfering with CPR because the probe is located in the esophagus. Owing to the increasing use of TEE during resuscitation (intra-arrest TEE), the American College of Emergency Physicians and the American Society of Echocardiography have jointly published guidelines for point-of-care applications in CA resuscitation [3]. Since the publication of these guidelines, considerable experience has been accumulated with TEE during CPR. Korea, one of the first countries to use TEE in the ED, began employing TEE accordingly in 1992 [4]. Recently, almost all EDs in Korea have been equipped with echocardiography machines; the remaining EDs are being prepared to support it. However, considering limitations in professional human resources and procedural time, it is unreasonable to conduct a comprehensive TEE study in the ED. By contrast, intra-arrest TEE as a point-of-care ultrasound is feasible in the ED and can be helpful in resuscitating patients who experienced CA. This review is intended to provide an overview of the practical use of intra-arrest TEE in emergency medicine.
INDICATIONS AND PREVIOUS EXPERIENCES
Intra-arrest TEE is performed to explore the mechanism of blood flow during CPR, diagnose the possible cause of CA, monitor the presence of cardiac contraction, assess the effectiveness of chest compression, guide catheter cannulation, and evaluate the complications of CPR. Table 1 summarizes the indications and objectives of intra-arrest TEE and the echocardiographic findings of intra-arrest TEE in the literature [5-47].
PREPARATION FOR INTRA-ARREST TEE
Multiple factors should be considered when implementing intraarrest TEE in the ED. These include the cost of the equipment; equipment maintenance, including probe disinfection; the input of additional personnel and technical training; evaluation of the operation quality; and collaboration with experts from other clinical fields, including cardiology [48]. For intra-arrest TEE, the TEE probe and related software must be purchased together with the echocardiography machine. The most appropriate area to place the echocardiography machine is a room in the resuscitation area. Drugs and devices for intensive monitoring and advanced life support, including airway support, defibrillation, and emergency medications, should be available at all times during TEE. According to Spaulding’s classification for disinfection and sterilization of patient care items and equipment, the TEE probe is classified as a semi-critical instrument with an endoscope. Cleaning and disinfection after every use are required according to these guidelines [49,50].
TEE should be performed by physicians who have (1) knowledge of cardiovascular anatomy and physiology, (2) knowledge of echocardiographic imaging, (3) proficiency and experience in performing TTE and TEE procedures, (4) knowledge required for interpretation of TEE results, and (5) knowledge of the management of TEE equipment and related instruments [51]. Considering these requirements, the guidelines recommend supervised performance and interpretation of at least 50 to 100 TEE procedures prior to the independent undertaking of TEE [52-54]. The American College of Emergency Physicians recommends a minimum of 10 proctored TEE examinations on live patients and simulation models with TEE-specific continuing medical education for the use of TEE in the ED for ultrasound-guided resuscitation during or after CA [55]. Therefore, emergency physicians performing TEE in the ED should have the qualifications for performing echocardiography, skills for TEE procedures, and competent TEE experience. Echocardiography is an operator-dependent procedure; therefore, the operator should conduct the procedure only after becoming certain that he or she can perform TEE on his or her own with consideration of the patient’s safety. Lack of substantial training may result in misdiagnosis or misinterpretation of echocardiographic findings, possibly causing catastrophic outcomes.
INTRA-ARREST TEE PROCEDURE
A physician who is capable of performing TEE and is not part of the CPR team is required for the intra-arrest TEE procedure. Intraarrest TEE is usually initiated after endotracheal intubation is complete. The patient in CA is not able to swallow the TEE probe; therefore, the operator must push the TEE probe into the esophagus. Before insertion of the TEE probe into the esophagus, the operator initially checks whether the probe is located behind the endotracheal tube and then bends the probe tip (anteflexion position) and pushes it into the back of the pharynx. When the TEE probe reaches the pharynx, it is inserted into the esophagus by straightening (unlocked position) and pushing the tip of the probe. If resistance is sensed while inserting the probe, the probe tip is not inserted into the esophagus. Insertion with excessive force may cause damage to the hypopharynx or upper esophagus [56,57]. Care should be taken not to dislodge the endotracheal tube during TEE.
IMAGING PROTOCOL AND VIEWS FOR INTRA-ARREST TEE
The protocol for intra-arrest TEE includes a quick scan to assess the possible cause of CA and the presence of cardiac contraction, assessment of CPR quality, monitoring of resuscitation measures, and guidance of catheter cannulation. To perform an initial scan immediately after probe insertion, the mid-esophageal (ME) fourchamber view, ME long-axis (ME LAX) view, ME ascending aorta short-axis (SAX) view, ME ascending aorta LAX view, descending aorta SAX view, and upper esophageal aortic arch LAX view are obtained in order (Fig. 1) [40]. Transgastric views during chest compressions are not recommend because the forceful anterograde or retrograde flexion position for the transgastric views may cause injury to the esophagus or stomach [58,59]. After quick observation of the heart and great vessels, a TEE probe with a four-chamber view is placed at the ME level to monitor for cardiac movement and assist resuscitation measures. When catheterization is required for resuscitative measures, such as employing a central venous catheter, extracorporeal membrane oxygenation, or resuscitative endovascular balloon occlusion of the aorta (REBOA), ME bicaval view and the descending aorta SAX and LAX view are optimal for visualizing the vena cava or aorta. After the return of spontaneous circulation, a comprehensive examination of cardiac function, morphology, and regional-wall motion is needed.

Suggested intra-arrest transesophageal echocardiography imaging planes and the corresponding structures or pathologies imaged. ME, mid-esophageal; RA, right atrium; RV, right ventricle; LA, left atrium; LV, left ventricle; MV, mitral valve; AV, aortic valve; TV, tricuspid valve; LVOT, left ventricular outflow tract; RVOT, right ventricular outflow tract; SVC, superior vena cava.
FINDINGS OF INTRA-ARREST TEE
The diagnostic sensitivity, specificity, and positive predictive value of TEE for the cause of CA were reported to be 93%, 50%, and 87%, respectively [17]. One-fourth of the patients who underwent intra-arrest TEE showed specific evidence associated with CA. Such evidence is associated with poor resuscitation outcome [40]. The common causes of CA confirmed by TEE are cardiac tamponade, aortic dissection, pulmonary embolism, and intracardiac thrombi. Cardiac tamponade is easily diagnosed and appears as a large amount of pericardial effusion with the collapse of the right ventricle (RV) in ME views (Fig. 2). A large pericardial effusion and/or left pleural effusion may be a sign of aortic rupture (Fig. 3). Aortic dissection can be diagnosed by the presence of an intimal flap in the ascending aorta, aortic arch, and descending aorta views (Fig. 4). High suspicion or diagnosis of pulmonary embolism is possible when thrombi are observed in the right atrium, RV, or pulmonary artery (Figs. 5, 6). Intracardiac or disseminated thrombi, observed as echogenic densities in the left-side cardiac chambers and/or the aorta without right heart thrombus, is a sequela of CA (Fig. 7). During a quick scan of the possible causes of CA, cardiac contraction and movement during chest compressions can be observed. Fibrillary or mechanical contractions of the ventricle can be observed during ventricular fibrillation or pulseless electrical activity (Supplemental Video 1). Catheterizations for interventional measures, such as REBOA or extracorporeal CPR, can be assisted by intra-arrest TEE [29,60]. Cardiac movement, including compression of the cardiac chambers and valvular motion, can be continuously monitored during TEE [7,8,14,21]. Kinetic analysis of chest compressions can be performed by measuring the excursions of the free wall of the RV [34]. An intracardiac shunt or paradoxical embolism can be detected during intra-arrest TEE [41,47].
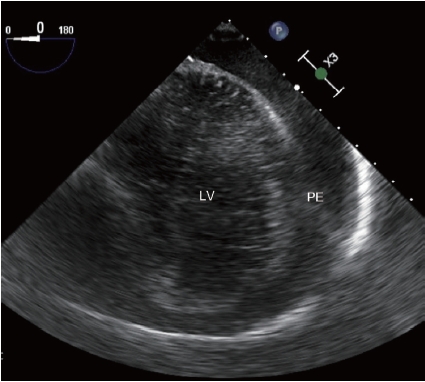
Cardiac tamponade. The mid-esophageal view shows a large amount of pericardial effusion (PE) surrounding the left ventricle (LV).
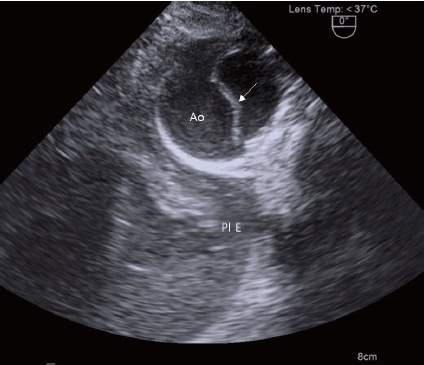
Ruptured aortic dissection. A dissecting flap (arrow) and left pleural effusion (Pl E) are noted on the descending aorta (AO) short-axis view.
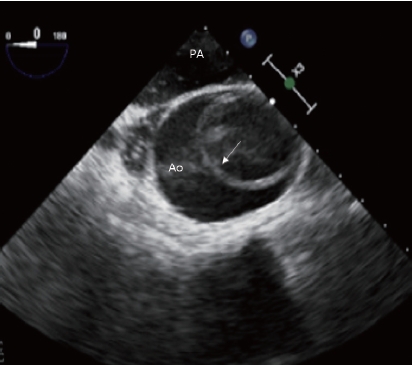
Aortic dissection. The mid-esophageal ascending aorta short-axis view shows a dissecting flap (arrow) in the ascending aorta (Ao). PA, pulmonary artery.

Thrombi (T*) in the right-sided chambers. Echogenic densities occupying the right atrium (RA) and the right ventricle (RV) on the mid-esophageal four-chamber view suggest pulmonary embolism as a possible cause of cardiac arrest. LA, left atrium; LV, left ventricle.

Thrombi (T*) in the pulmonary artery (PA). The mid-esophageal view for the PA shows echogenic densities in the main and right PA. Ao, ascending aorta.
SAFETY OF INTRA-ARREST TEE
TEE is relatively safe. The overall complication rate of diagnostic and intraoperative TEE ranges from 0.18% to 2.8%, and the mortality rate is less than 0.01% to 0.02% [61-63]. However, no study has reported the safety of intra-arrest TEE. From our experience (unpublished data), intra-arrest TEE was successfully performed with no complications in 179 of 183 patients (97.8%) who experienced out-of-hospital CA. Failure of TEE probe insertion into the esophagus occurred in three patients (1.6%), while vallecular injury was confirmed in one patient (0.5%) after intra-arrest TEE. The electrical safety of using TEE during transthoracic defibrillation remains controversial [64]. Operator injury or equipment failure was not reported during transthoracic defibrillation. No study has reported the harmful effects of transthoracic defibrillation on operators or patients when a TEE probe is inserted. However, the effect of defibrillation on TEE machines has not yet been evaluated.
CONCLUSION
TEE is now widely practiced not only by cardiologists, but also by doctors who manage patients with cardiovascular disorders, including emergency physicians, intensivists, and anesthesiologists. Excellent imaging windows, easy accessibility, and high portability have enabled TEE to be a point-of-care imaging modality during CA in the ED. Intra-arrest TEE facilitates the diagnosis of the CA cause, enables the evaluation of cardiac contractions and CPR quality, and aids in catheter insertion for therapeutic procedures. Trained experts, protocols, coordination, and equipment maintenance are essential for the successful application of an intra-arrest TEE program in the ED. Future research should evaluate the effect of intra-arrest TEE on resuscitation outcomes in patients with CA.
SUPPLEMENTARY MATERIAL
Supplementary material is available at https://doi.org/10.15441/ceem.22.399.
Supplemental Video 1.
Illustrative case of ventricular fibrillation observed on intra-arrest transesophageal echocardiography. The mid-esophageal four-chamber view shows fine fibrillary contraction of the left ventricle and mitral valve movement.
Notes
CONFLICT OF INTEREST
No potential conflict of interest relevant to this article was reported.
FUNDING
None.
AUTHOR CONTRIBUTIONS
Conceptualization: SOH; Data curation: WJJ; Investigation: KCC; Methodology: SOH; Resources: WJJ; Supervision: SOH; Visualization: YIR; Writing–original draft: SOH; Writing–review & editing: all authors.
All authors read and approved the final manuscript.
References
Article information Continued
Notes
Capsule Summary
What is already known
The use of point-of-care ultrasound during cardiopulmonary resuscitation (CPR) has been suggested.
What is new in the current study
Intra-arrest transesophageal echocardiography can be used as a point-of-care ultrasound method to diagnose the cause of cardiac arrest, determine the presence of cardiac contractions, evaluate the quality of CPR, assist with catheter insertion, and explore the mechanism of blood flow during CPR.