INTRODUCTION
Brown-Séquard syndrome (BSS) is a neurological condition associated with trauma-related spinal cord injury. This entity was first reported by Charles-Edouard Brown-Séquard in the 1840s. BSS involves ipsilateral hemiplegia and contralateral loss of both pain and temperature sensation below the level of the lesion, after spinal cord hemisection.
Acute-onset BSS is frequently associated with a penetrating injury, whereas late-onset BSS is extremely rare and develops weeks to years after a blunt injury [1-3]. The prognosis of blunt injury-associated BSS is usually good. To our knowledge, however, acute-onset BSS following blunt injury has rarely been reported. Here, we describe a case of BSS that developed immediately after a blunt injury, presenting with unilateral loss of thoracic motion.
CASE REPORT
A previously healthy 58-year-old man was transferred to our emergency department (ED) following a motor vehicle accident, in which his car rolled over after unintentionally hitting a guardrail. At the time of the accident, he was driving without wearing a seat belt and the airbag was not deployed. Immediately after the accident, he experienced neck pain and dyspnea accompanied by complete paralysis of all extremities. However, during transportation, his left paresis started to resolve, and showed complete resolution by the time of arrival at the ED.
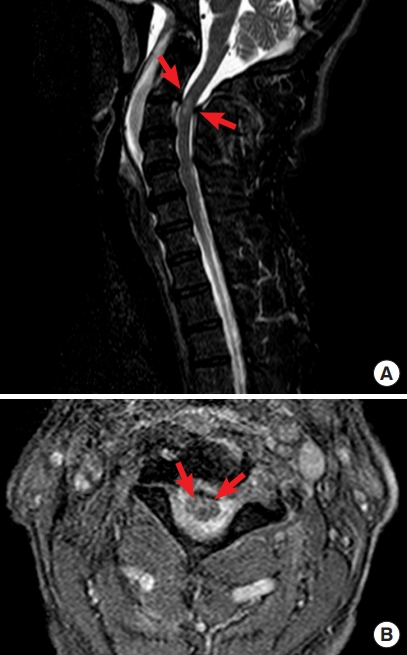
On ED arrival, the patient reported experiencing dyspnea, pain in the back of the neck, and right hemiparesis. Vital signs were as follows: blood pressure, 128/78 mmHg; heart rate, 72 beats/min; respiratory rate, 28 breaths/min with a peripheral capillary oxygen saturation of 98% on ambient air; and body temperature, 35.3°C. His mental status appeared slightly depressed, with a Glasgow Coma Scale score of 14 (E3 V5 M6). The back of his neck was tender, without apparent evidence of trauma. Loss of thoracic motion was observed on the right side. Neurological examination revealed complete right paresis with a positive Babinski reflex on the right side. Pain and temperature sensations were lost below the C3 level on the left side. Urinary retention and proctoparalysis were also noted. Given the distribution of his neurological abnormalities, a presumptive diagnosis of BSS as a manifestation of cervical spinal cord injury was made. Chest radiography showed that his right diaphragm was at the level of the ninth rib posteriorly, two intercostal spaces higher than the left diaphragm (Fig. 1), suggestive of right-sided diaphragmatic elevation due to right phrenic nerve paralysis. Brain and cervical computed tomography did not show any cervical fractures or vertebral artery dissection. However, cervical magnetic resonance imaging revealed hyperintensities within the cervical spinal cord at the C2-C3 level, indicating spinal cord injury without computed tomography evidence of trauma (SCIWOCTET), and therefore, confirming our clinical diagnosis (Fig. 2).
The patient was admitted to the intensive care unit for close observation because of concerns that the unilateral loss of thoracic motion and spinal injury at a high cervical level could lead to respiratory compromise. Normal motion of his right thorax resumed on the third day. The motor deficit owing to right paresis was initially classified, based on the American Spinal Injury Association (ASIA) impairment classification [4], as ASIA C. His right paresis started to improve on the eighth day. No respiratory or circulatory compromise developed during hospitalization. The patient was gradually able to walk with cane support; however, he had an intermittent need for self-catheterization and laxative use to help with urinary retention and lack of bowel movement, respectively. He was finally transferred to a rehabilitation facility after 41 days of hospitalization. At his 3-year follow-up appointment, genitourinary and gastrointestinal symptoms, as well as hemiparesis (ASIA D) were still present.
DISCUSSION
The etiology of BSS involves trauma, being associated with 1% to 4% of spinal cord injuries [5]. Acute-onset BSS frequently occurs after a penetrating injury, whereas late-onset BSS is rare and is associated with a blunt injury, developing after 6 weeks to 6 years [1-3]. In general, injury-associated BSS has a good prognosis [1-3]; 75% to 90% of patients regain the ability to walk independently by the time of discharge from rehabilitation [5]. However, BSS associated with blunt injury has a better prognosis than that associated with penetrating injury [2,3]. Acute-onset BSS following blunt injury is rare, and the findings of a case series of ten patients with BSS following blunt injury suggested that neurological impairment is significantly reduced after the initial clinical evaluation [6]. In contrast, our patient, who had a blunt injury-associated BSS, showed acute-onset symptoms with a poor prognosis.
SCIWOCTET, a spinal cord injury without radiographic abnormality-like condition, is rare with a reported prevalence of 3% among adult patients with injury in the cervical region [7]. To our knowledge, only a few cases of BSS have been associated with SCIWOCTET, and they developed immediately after a blunt trauma [8-10], similar to what happened in our case. However, in contrast to ours, their cases reportedly showed short-term neurological improvements.
In a healthy individual, the dome of the right diaphragm is positioned at the level of the fifth rib anteriorly and at the level of the tenth rib posteriorly. A diagnosis of right diaphragm paralysis is established when the distance between the right and the left diaphragm is greater than two intercostal spaces on chest radiography [11]. Lung ultrasonography can also aid in establishing this diagnosis based on the thickness of the diaphragm or the change in its position between inspiration and expiration. In the case presented here, the diagnosis of right diaphragm paralysis was definitive. Although we did not track the position of the patient’s right diaphragm, we found, on the chest radiograph obtained on admission, that it was at the level of ninth rib posteriorly and two intercostal spaces higher than the left diaphragm. Additionally, our patient had an uneventful clinical course with respect to respiration: his right thoracic motion improved. For this reason, follow-up chest radiography or other diagnostic modalities were not pursued.
The phrenic nerve is derived from the spinal anterior horn cells between the C3 and C5 levels. Due to the injury at the C3 level, which affected the associated phrenic nerve, our patient had a unilateral loss of thoracic motion. Thus, the absence of unilateral thoracic motion can be a sign of BSS, and clinicians should highly suspect injuries to the cervical spine level in patients with a unilateral loss of thoracic motion.
Published studies suggest that blunt injury-associated BSS is rare with late-onset symptoms. Nonetheless, BSS as a manifestation of cervical cord injury should be highly suspected when trauma patients present with hemiparesis and contralateral sensory loss that develops immediately after trauma. Furthermore, unilateral loss of thoracic motion can be a sign of BSS. Patients and clinicians should be informed of the potential poor prognosis of blunt injury-associated BSS.