AbstractObjectiveA cardiothoracic ratio Ōēź0.50 is widely used as an indicator of cardiomegaly, but associations between the cardiothoracic ratio and left ventricular systolic dysfunction (LVSD) have not been investigated previously. We conducted this study to investigate the relationship between cardiothoracic ratio measured using computed tomography (CT) and left ventricular ejection fraction (LVEF), and to determine the optimal cardiothoracic ratio for predicting left ventricular systolic dysfunction (LVSD).
MethodsA retrospective cross-sectional study was performed using data from patients who underwent both chest CT and echocardiography at the emergency department from January 1 to December 31, 2021. The patients were classified as normal, or having mild, moderate, and severe LVSD based on their LVEF, and the cardiothoracic ratios of each group were compared. The receiver operating characteristic (ROC) curve analyses were used to identify the optimal cardiothoracic ratio for prediction of mild, moderate, and severe LVSD.
ResultsThe final study population included 444 patients. The median CT-measured cardiothoracic ratio was 0.54 for patients with normal LVEF, and 0.60 for patients with LVSD (P<0.001). The optimal CT-measured cardiothoracic ratios for predicting mild, moderate, and severe LVSD were 0.56, 0.59, and 0.60, and their areas under the ROC curve were 0.653, 0.690, and 0.680, and negative predictive values were 90%, 94%, and 98%, respectively.
ConclusionThe best cutoff value for a CT-measured cardiothoracic ratio suggestive of LVSD was 0.56, which is very different from the 0.50 value typically considered an abnormal cardiothoracic ratio. The CT-measured cardiothoracic ratio Ōēź0.56 can be used as a rough indicator of mild LVSD, and a ratio <0.60 can exclude severe LVSD with a high degree of confidence.
INTRODUCTIONFirst described in 1919, the cardiothoracic ratio is the most widely used classic indicator of cardiomegaly on chest radiographs [1]. The cardiothoracic ratio is the ratio of the maximum horizontal heart diameter to the maximum horizontal chest diameter measured in the posteroanterior (PA) thoracic radiograph and is calculated using up to two decimal places. A cardiothoracic ratio of 0.42 to 0.50 in PA thoracic radiographs is considered normal. Previous studies have reported that an elevated cardiothoracic ratio (>0.50) is significant for prognostic assessment in healthy adults [2], the elderly [3], and patients with various congenital [4] and acquired heart diseases [5ŌĆō10]. In addition, Shah et al. [11] reported that a cardiothoracic ratio greater than 0.50 was a good predictor of decreased left ventricular systolic function. In addition, in a group of hemodialysis patients, cardiothoracic ratios greater than 0.55 were found to be the most important independent prognostic factor associated with all-cause mortality within 2 years [12], and in patients undergoing valve replacement, a cardiothoracic ratio greater than 0.60 is known to be an independent prognostic factor associated with death in the 1st year after valve replacement [13].
The cardiothoracic ratio should not be measured on anteroposterior (AP) chest radiographs because the heart shadow is artificially enlarged due to the divergence of the X-ray beam. However, chest radiography is often performed using the AP view rather than the PA view for patients with impaired mobility, making it impossible to measure the cardiothoracic ratio. Kim et al. [14] reported that the cardiothoracic ratio can easily be obtained by measuring the maximum heart width divided by the maximum chest width in single computed tomography (CT) images and can predict the presence of a cardiomegaly if the cardiothoracic ratio exceeds 0.50. CT is performed very frequently in the emergency department (ED), and when CT is performed, the cardiothoracic ratio can be measured by CT instead of chest radiography. We conducted this study to investigate the associations between the cardiothoracic ratio measured by chest CT and left ventricular systolic function measured by echocardiography, and to determine the best cardiothoracic ratio to predict left ventricular systolic dysfunction (LVSD).
METHODSEthical statementsThis study was implemented after approval by the Institutional Review Board of Kangbuk Samsung Hospital (No. 2022-06-058). The Institutional Review Board exempted written informed consent due to the retrospective nature of the study. Personal information, such as patient name, date of birth, and social identification number, was deleted after assigning research subject numbers to ensure anonymity. This study was conducted in compliance with the World Medical Association Declaration of Helsinki [15].
Study design and subjectsA retrospective cross-sectional study of adult patients over the age of 18 years was conducted. The study included patients who underwent both (contrast or noncontrast enhancement) chest CT and echocardiography in the ED. The exclusion criteria were (1) an interval of more than 24 hours between CT and echocardiography imaging (given left ventricular ejection fraction [LVEF] may change over time if there is a change in systemic vascular resistance or effective circulating volume, or if the heart becomes hyperdynamic); (2) no LVEF report on echocardiography; and (3) anatomical abnormalities rendering heart or chest transverse diameter measurement on CT images impossible (Fig. 1).
The sample size for multivariate logistic regression was calculated using the following formula based on PeduzziŌĆÖs research: N=10├Śk/p, where k is the number of independent variables and p is the smallest of the proportions of negative or positive cases in the population [16]. When calculated using the above method, and assuming five independent variables to be included in the analyses, and that the probability of a case of reduced left ventricular systolic function (LVEF<50%) based on the results of echocardiography herein is 18%, the minimum sample size required was determined to be 278 cases. To satisfy the minimum sample size, the retrospective study period was set to the past 1 year.
Outcome measuresDuring the study, CT scans were performed using a Brilliance iCT-SP 128 CT scanner (Philips Medical Systems) in accordance with the protocol of the radiology department. The default values for the recombinant parameters used were a 2-mm slice thickness and 2-mm slice spacing. The measurements were performed using the length measurement tool built into the INFINITT PACS viewer (INFINITT Healthcare) program in a 27-inch FHD (1,920├Ś1,080) resolution monitor environment. To select an axial image with maximal cardiac width, the axial images were continuously moved on the mediastinum window setting, cross-referencing the mid-sagittal image on a 1├Ś2-split screen. The maximum heart width was measured in the selected axial image and the maximum chest width was measured in the same image. If there was a shadow presumed to be pericardial effusion or pericardial fat, this shadow was included in the measurement of the maximal cardiac width. The maximum chest width was measured between the inner edges of the ribs. In this way, the CT cardiothoracic ratio was calculated as the value obtained by dividing the maximum heart width by the chest width in the same image (Fig. 2). The cardiothoracic ratio was measured by three emergency physicians and the interobserver agreement between the observers was assessed using the intraclass correlation coefficient (ICC). The ICC and the 95% confidence interval (CI) were calculated by the two-two-way random-effects model in condition of absolute agreement (k=3).
LVEF was retrieved from the final echocardiography report. During the study period, echocardiography was performed by certified sonographers and the final report was confirmed by the attending cardiologist. Either the Teichholz or modified Simpson method was used to measure the ejection fraction. The Teichholz method was adopted for patients who did not have regional left ventricular wall motion abnormalities (RWMA) and the modified Simpson method was adopted for patients with RWMA. Normal left ventricular systolic function was defined as an LVEF of 50% or more, mild LVSD was defined as an LVEF of 40% to 49%, moderate LVSD was defined as an LVEF of 30% to 39%, and severe LVSD was defined as an LVEF less than 30%.
Statistical analysesAll statistical analyses were performed using Stata ver. 15.1 (Stata Corp). Continuous variables were analyzed using a nonparametric method (Mann-Whitney U-test) because they did not follow a normal distribution, and representative values were presented as medians and interquartile ranges. Nominal variables were analyzed using a chi-square test and presented as frequencies and percentages. A P-value of less than 0.05 was considered to statistically significant. A logistic regression analysis was performed to analyze whether the classic cardiothoracic ratio of 0.5 or higher is correlated with LVSD. Comparison of the cardiothoracic ratio according to the degree of LVSD (normal, mild, moderate, and severe LVSD) was performed using a one-way analysis of variance. Using receiver operating characteristic (ROC) curve analyses, the optimal cutoff values of the cardiothoracic ratio to predict mild, moderate, and severe LVSD were calculated, and the sensitivity and specificity of each cutoff value were calculated. We interpreted area under the ROC curve (AUC) values between 0.9 and 1.0 as being excellent, 0.8 to 0.9 as good, 0.7 to 0.8 as fair, 0.6 to 0.7 as poor, and 0.5 to 0.6 as failures.
RESULTSThe study period was 1 year from January 1, 2021 to December 31, 2021. During the study period, 1,119 adult patients underwent both (noncontrast or contrast-enhanced) chest CT and echocardiography in our ED. Of these 1,119 patients, 672 were excluded because they underwent echocardiography 24 hours after CT, and another three patients were excluded because their echocardiography report did not describe the LVEF. Finally, 444 patients were selected as study subjects (Fig. 1). The reasons for echocardiography were to rule out heart failure (39.2%), rule out ischemic heart disease (28.4%), rule out cor pulmonale (6.3%), rule out aortic disease 6.1%, fever workup (5.6%), rule out pericardial disease (4.1%), rule out valvular heart disease (3.8%), rule out cardiomyopathy (3.4%), chest trauma (1.8%), postcardiac-arrest workup (0.9%), and preoperative check (0.5%). There were no patients with anatomical deformities severe enough to prevent measurement of the cardiothoracic ratio.
The degree of consistency between the three measurers of the cardiothoracic ratio measurement was excellent. The ICC for the maximum heart width measurement was 0.959 (95% CI, 0.909ŌĆō0.981; P<0.001), and the ICC for the maximum chest width measurement was 0.937 (95% CI, 0.885ŌĆō0.967; P<0.001).
The general characteristics of the study subjects are presented in Table 1. The mean age of patients who underwent both chest CT and echocardiography within 24 hours in the ED was 78 years. Among all study subjects, 18.7% had LVSD. The median cardiothoracic ratio of patients with LVSD was 0.60, which was significantly different from the median cardiothoracic ratio of 0.54 in patients with normal left ventricular function (Table 1).
The median cardiothoracic ratio according to the degree of LVSD is presented in Table 2. Of the 444 total subjects, 81.3% had normal left ventricular systolic function, and their median cardiothoracic ratio was 0.54. The median cardiothoracic ratios of the mild, moderate, and severe LVSD groups were 0.57, 0.60, and 0.61, respectively. The cardiothoracic ratios differed significantly between the normal group and the moderate or severe LVSD groups, but not between the normal group and the mild LVSD group (Table 2).
When applying the traditional abnormal cardiothoracic ratio threshold of 0.50, the odds ratio for LVSD (LVEF<50%) was only 1.95 and there was no significant difference (Table 3).
The optimal cardiothoracic ratios for prediction of mild, moderate, and severe LVSD were 0.56, 0.59, and 0.60, respectively. The AUC of the 0.56 cardiothoracic ratio cutoff value for mild LVSD was 0.653, and was 0.690 for the 0.59 cutoff for moderate LVSD, and 0.680 for the 0.60 cutoff for severe LVSD. There was no cardiothoracic ratio criterion with an AUC of 0.700 or more (Table 4).
The positive predictive value (PPV) of the cardiothoracic ratio to predict LVSD was less than 30%, and the negative predictive value (NPV) was very high (Ōēź90%) (Table 4). Only 23 patients (5.18%) had LVSD despite having cardiothoracic ratios of 0.56 or less, while 18 patients (4.1%) had moderate or severe LVSD despite having cardiothoracic ratios of 0.59 or less, and nine patients (2.0%) had severe LVSD despite cardiothoracic ratios of 0.60 or less. There were four atypical patients who had a LVEF of <40% even though they had a cardiothoracic ratio of <0.5 (Fig. 3).
DISCUSSIONThe results of this study showed that the median cardiothoracic ratio in the normal left ventricular systolic function group was 0.54 (interquartile range, 0.50ŌĆō0.60), and that the traditional abnormal cardiothoracic ratio standard of 0.50 cannot be utilized to identify impaired ventricular systolic function. A cardiothoracic ratio of 0.50 is typically considered suggestive of cardiomegaly, but our findings indicate the cardiothoracic ratio criterion for prediction of LVSD could be set to 0.56 or greater; however, the AUC for the 0.56 value was less than 0.700. The optimal cardiothoracic ratio presented in this study is therefore not a decisive or absolute standard, but rather a rough indicator of LVSD to be used in conjunction with other diagnostic findings.
To the best of our knowledge, there have been few studies assessing the predictive value of the CT-measured cardiothoracic ratio when diagnosing LVSD. A recent study by Kaiume et al. [17] is especially noteworthy. They reported that the maximum transverse cardiac diameter and CT ratio are useful for detecting LVSD, with AUCs of 0.794 and 0.746, respectively. The Kaiume et al. [17] results are consistent with our finding that the CT-measured cardiothoracic ratio can predict reductions in left ventricular systolic function. Major advantages of the current study as compared with Kaiume et al. [17] are a larger number of patients (83 vs. 39 patients with LVSD), and shorter interval between echocardiography and CT imaging (less than 24 hours vs. 3 months).
The optimal CT-measured cardiothoracic ratios for prediction of mild, moderate, and severe LVSD were found to be 0.56, 0.59, and 0.60, respectively. It is particularly noteworthy that the NPV of the cardiothoracic ratio for LVSD is very high: cardiothoracic ratios of <0.56, <0.59, and <0.60 can predict the absence of LVSD with 90%, 94%, and 98%, respectively. In patients with LVSD, the decision to administer a bolus of intravenous fluids or several important medications with negative inotropic effect (such as ╬▓-blockers or calcium channel blockers) should be made judiciously. The high NPV of the cardiothoracic ratio in predicting systolic dysfunction may improve patient care when echocardiography is not immediately available but intravenous fluid or medications with negative inotropic effect are necessary.
The results of this study reaffirmed that it is difficult to accurately predict left ventricular systolic function using the cardiothoracic ratio. Although the moderate and severe LVSD groups had significantly greater cardiothoracic ratios than that of the normal systolic function group, the AUC was not greater than 0.700 no matter which cardiothoracic ratio criteria were used. This result was consistent with the results of the Philbin et al. [18] study that reported the chest radiographic cardiothoracic ratio and LVEF were only slightly negatively correlated, making it difficult to predict LVEF accurately using the cardiothoracic ratio alone, and measurement of left ventricular function required direct measurement using echocardiography. Nonetheless, echocardiography is a labor-intensive test that takes a considerable amount of time even when performed by a skilled expert, and is almost impossible to have performed at night or on holidays. In normal patients, the test time is about 10 to 20 minutes, but if abnormalities are found, echocardiographic scans can take as long as 1 hour [19]. The growing demand for echocardiography has led to wait times of several days even in developed countries. A study in the United Kingdom found that the median wait time for inpatients to undergo an echocardiography was 2 days, the median wait time for outpatients was 8 weeks, and the median waiting time was more than 4 months in 21% of cases [20]. In our hospital, echocardiography is only available during the daytime, and the waiting time for outpatient echocardiography is 7 days. For ED patients, echocardiography is usually done on the same day, but recently the number of cases for whom echocardiography cannot be performed on the day of visit is increasing. Therefore, even though the cardiothoracic ratio cannot replace echocardiography for definitive measurement of the LVEF, the cardiothoracic ratio can be used as a rough indicator of LVSD when echocardiography is not available.
Wingate-Saul et al. [21] reported that when echocardiography is performed based on an increased cardiothoracic ratio, the positive prediction is only 6%. The results of the current study also reaffirmed that the PPV of cardiothoracic ratio in predicting LVSD is low, and no matter what cutoff value was used, the PPV did not exceed 30%. Increased cardiothoracic ratio should not be used as a sole indication for echocardiography.
The limitations of this study are as follows. First, selection bias likely occurred in the process of selecting only patients who had undergone both chest CT and echocardiography in the ED. In fact, the patients selected for this study were mostly elderly patients. Therefore, the results of this study cannot be extended to the young patient group. Second, the interobserver agreement between echocardiographers for LVEF measurement was not assessed. Third, the presence of underlying diseases that can affect the measurement of the cardiothoracic ratio, such as chronic obstructive pulmonary disease, heart failure, previous heart surgery, and use of medications that can affect the LVEF, were not investigated due to the retrospective nature of this study. When inotropic agents have been used, the LVEF is typically enhanced among patients with an elevated cardiothoracic ratio. Therefore, if use of inotropic agents had been investigated and adjusted for in the analyses, the study results would have been more robust. Fourth, although the simplest and most widely used method to investigate LV contractility is measuring LVEF by echocardiography, low ejection fraction and LVSD are not equivalent if there is a change in systemic vascular resistance or effective circulating volume, or if the heart becomes hyperdynamic. However, more accurate methods such as nuclear medicine tests or cardiac magnetic resonance imaging were not performed for our study subjects. In this study, we had no choice but to use the LVEF as a means of estimating LVSD. Lastly, the cardiothoracic ratios reported in this study were measured using CT and therefore cannot be applied to cardiothoracic ratio measured using chest radiographs.
In conclusion, there is a significant association between the CT-measured cardiothoracic ratio and LVSD. The optimal cutoff value for CT-measured cardiothoracic ratios suggestive of LVSD was 0.56, which is very different from the traditionally defined of abnormal cardiothoracic ratio of 0.50. The optimal CT-measured cardiothoracic ratios for prediction of mild, moderate, and severe LVSD were 0.56, 0.59, and 0.60, respectively. We suggest a CT-measured cardiothoracic ratio of >0.56 can be used as a rough indicator of mild LVSD. The CT-measured cardiothoracic ratio had a high NPV for LVSD, and a CT-measured cardiothoracic ratio of <0.60 can exclude severe LVSD with a high degree of confidence.
REFERENCES1. Danzer CS. The cardiothoracic ratio: an index of cardiac enlargement. Am J Med Sci 1919; 157:513-21.
2. Hemingway H, Shipley M, Christie D, Marmot M. Is cardiothoracic ratio in healthy middle aged men an independent predictor of coronary heart disease mortality? Whitehall study 25 year follow up. BMJ 1998; 316:1353-4.
3. Frishman WH, Nadelmann J, Ooi WL, et al. Cardiomegaly on chest X-ray: prognostic implications from a ten-year cohort study of elderly subjects: a report from the Bronx Longitudinal Aging Study. Am Heart J 1992; 124:1026-30.
4. Dimopoulos K, Giannakoulas G, Bendayan I, et al. Cardiothoracic ratio from postero-anterior chest radiographs: a simple, reproducible and independent marker of disease severity and outcome in adults with congenital heart disease. Int J Cardiol 2013; 166:453-7.
5. Hammermeister KE, Chikos PM, Fisher L, Dodge HT. Relationship of cardiothoracic ratio and plain film heart volume to late survival. Circulation 1979; 59:89-95.
6. Zaman MJ, Sanders J, Crook AM, et al. Cardiothoracic ratio within the ŌĆ£normalŌĆØ range independently predicts mortality in patients undergoing coronary angiography. Heart 2007; 93:491-4.
7. Cohn JN, Johnson GR, Shabetai R, et al. Ejection fraction, peak exercise oxygen consumption, cardiothoracic ratio, ventricular arrhythmias, and plasma norepinephrine as determinants of prognosis in heart failure. The V-HeFT VA Cooperative Studies Group. Circulation 1993; 87(6 Suppl):VI5-16.
8. Giamouzis G, Sui X, Love TE, Butler J, Young JB, Ahmed A. A propensity-matched study of the association of cardiothoracic ratio with morbidity and mortality in chronic heart failure. Am J Cardiol 2008; 101:343-7.
9. Ernst ER, Shub C, Bailey KR, Brown LR, Redfield MM. Radiographic measurements of cardiac size as predictors of outcome in patients with dilated cardiomyopathy. J Card Fail 2001; 7:13-20.
10. Berman M, Aravot D, Ben-Gal T, Sahar G, Sagie A, Vidne B. Cardiothoracic ratio: important prognostic tool in heart failure patients who are candidates for heart transplantation. Transplant Proc 2000; 32:727-8.
11. Shah S, Davies MK, Cartwright D, Nightingale P. Management of chronic heart failure in the community: role of a hospital based open access heart failure service. Heart 2004; 90:755-9.
12. Ito K, Ookawara S, Ueda Y, et al. A higher cardiothoracic ratio is associated with 2-year mortality after hemodialysis initiation. Nephron Extra 2015; 5:100-10.
13. Jiang L, Chen WG, Geng QS, et al. The cardiothoracic ratio: a neglected preoperative risk-stratified method for patients with rheumatic heart disease undergoing valve replacement surgery. Eur J Cardiothorac Surg 2019; 55:511-7.
14. Kim YS, Park HJ, Park SH, Chun HJ, Choi BG. A CT criteria of cardiomegaly. J Korean Radiol Soc 2007; 57:235-8.
15. World Medical Association declaration of Helsinki. Recommendations guiding physicians in biomedical research involving human subjects. JAMA 1997; 277:925-6.
16. Peduzzi P, Concato J, Kemper E, Holford TR, Feinstein AR. A simulation study of the number of events per variable in logistic regression analysis. J Clin Epidemiol 1996; 49:1373-9.
17. Kaiume M, Kurokawa R, Maeda E, Daimon M, Abe O. Detection of left ventricular dysfunction on axial non-contrast chest CT. Eur J Radiol 2022; 150:110274.
18. Philbin EF, Garg R, Danisa K, et al. The relationship between cardiothoracic ratio and left ventricular ejection fraction in congestive heart failure. Arch Intern Med 1998; 158:501-6.
19. Kang G. Service operations strategy of tertiary hospitals for medical supply chain improvement [masterŌĆÖs thesis]. Korea Advanced Institute of Science and Technology; 2004.
20. Macdonald MR, Hawkins NM, Balmain S, Dalzell J, McMurray JJ, Petrie MC. Transthoracic echocardiography: a survey of current practice in the UK. QJM 2008; 101:345-9.
21. Wingate-Saul L, Javaid Y, Chambers J. Echocardiography is not indicated for an enlarged cardiothoracic ratio. Br J Cardiol 2013; 20:149-50.
Fig.┬Ā1.Flow diagram for the study. ED, emergency department; CT, computed tomography; LVEF, left ventricular ejection fraction. 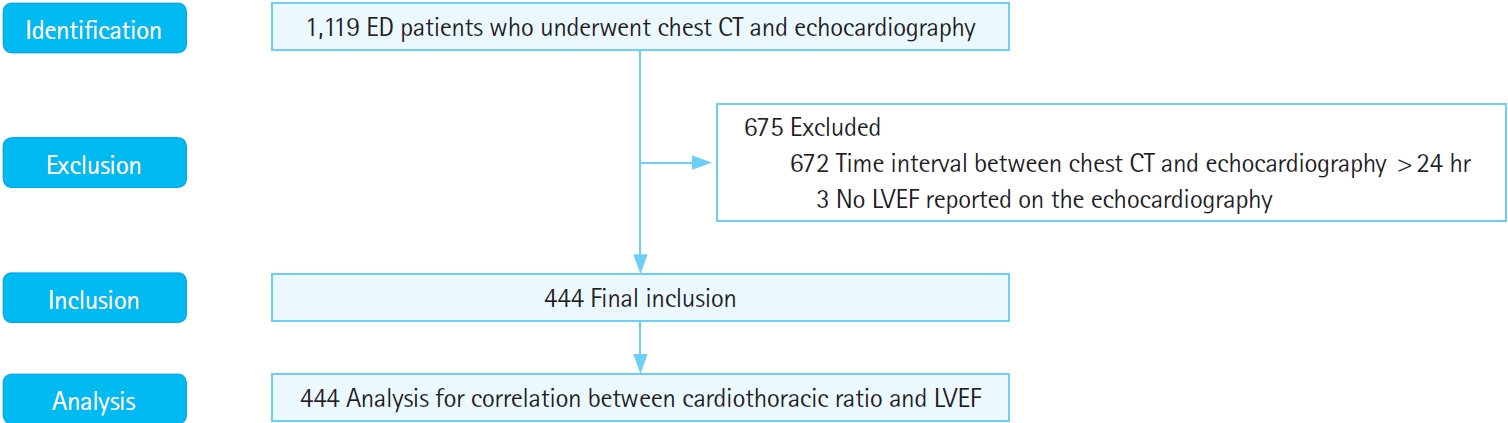
Fig.┬Ā2.Measuring the cardiothoracic ratio on computed tomography (CT). The cardiothoracic ratio was calculated by measuring the diameter of the heart (solid line) and the diameter of the chest (dotted line) on the axial CT image where the transverse diameter of the heart appeared widest. The measurements were obtained on the same basis for both (A) noncontrast CT and (B) contrast-enhanced CT images. 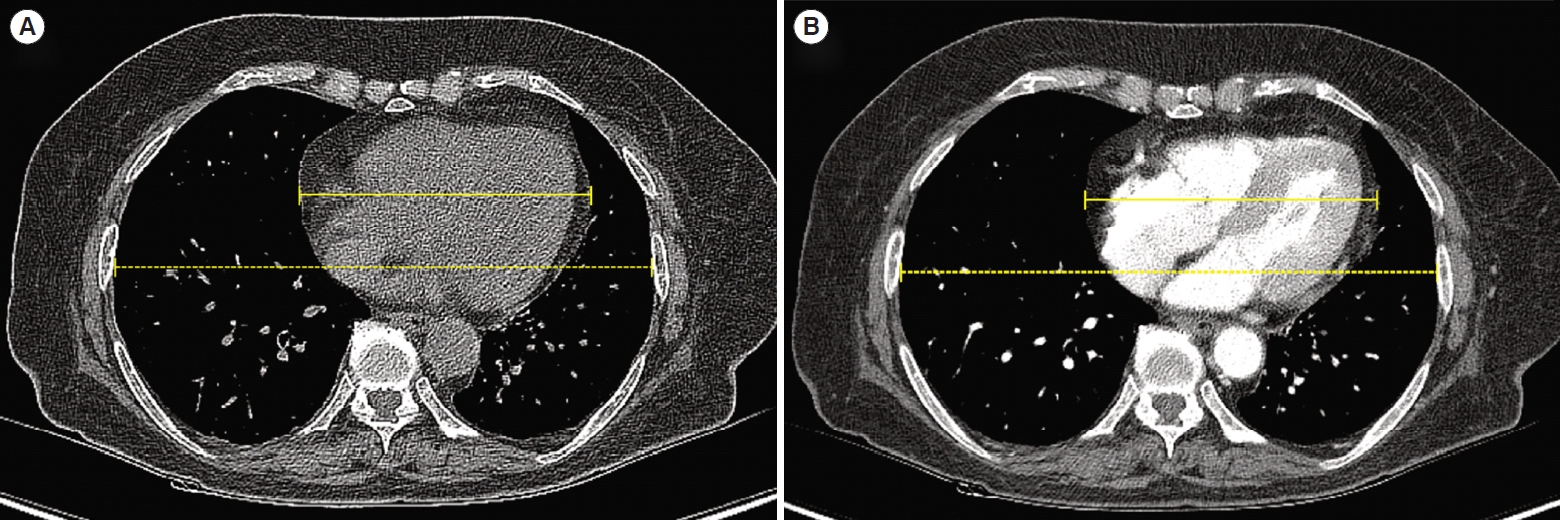
Fig.┬Ā3.A scatter plot of left ventricular ejection fraction (LVEF) according to the cardiothoracic ratio. The LVEF and the cardiothoracic ratio show a weak negative correlation. White circles indicate four atypical patients with an LVEF of less than 40 mmHg without cardiomegaly. 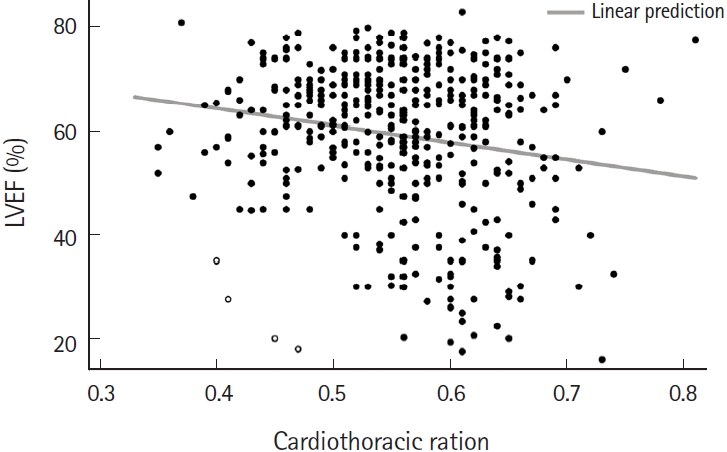
Table┬Ā1.General characteristics of the study patients Table┬Ā2.Cardiothoracic ratios measured by computed tomography according to LVEF (n=444)
Table┬Ā3.Predictive value of a cardiothoracic ratio greater than the 0.50 standard to identify a left ventricular ejection fraction less than 50%
Table┬Ā4.Optimal cardiothoracic ratio measured by computed tomography to predict LVSD |
|
|||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||