AbstractThe current heightened international political climate is accompanied by increased risk of chemical or biological agent weaponization. Historical accounts of biochemical warfare are extensive, and considering the recent use of such agents for targeted attacks, clinicians need to recognize and manage these cases. However, agent properties such as the color, odor, ability to be aerosolized, and long incubation period can introduce difficulties in the diagnostic and management approach. We searched PubMed and Scopus for a colorless, odorless, aerosolized substance with an incubation period of at least 4 hours. Data from articles were summarized and reported by agent. Based on data from the available literature, we included agents such as nerve agents, ricin, botulinum toxin, anthrax, tularemia, and psittacosis in this review. We also highlighted potential chemical and biological agents that could be weaponized and the optimal strategies for the diagnosis and treatment of patients exposed to an unknown aerosolized biological or chemical bioterrorism agent.
INTRODUCTIONIn the current international political climate, the risk of chemical or biological agent weaponization is undeniably heightened [1–3]. The 1972 Biological Weapons Convention and the 1993 Chemical Weapons Convention established safeguards to limit the use of biological and chemical weapons [4,5]. However, terrorist groups, rogue governmental agencies, or malicious individuals with access to these substances may elect to use the agents at any time. The COVID-19 pandemic has exposed the vulnerability of the American healthcare system to a biological agent [6]. Easily accessible information around advancements in microbiology, genetic engineering technology, and gene editing tools exacerbate the risk of a potential attack [7].
The use of biological agents for bioterrorism warfare extends from the ancient times to the present day; historical accounts document attackers throwing plague corpses over the city walls of Caffa (now Feodosia, Ukraine) during a siege [8]. Other accounts describe poisoning enemy wells with human remains in wartime [9]. Notable examples from the last century include the use of plague, cholera, and typhoid by the Japanese against China during World War II and the use of biological toxins by Iraq against ethnic Kurds and Iran in the 1990s [10,11]. Beyond open warfare, terrorists have used these agents to launch domestic attacks. The most prominent was the 2001 anthrax attacks in which an unknown perpetrator sent Bacillus anthracis spores to government officials and private citizens through the US Postal Service [12].
While biological agents are derived from naturally occurring organisms, their chemical counterparts are synthetically produced. The Tokyo subway sarin attack and the use of VX agent in the assassination of Kim Jong-nam have demonstrated the devastation potential of chemical weapons [13,14]. These agents are more common due to their availability and physiologic properties [15]. Unlike biological agents, victims of chemical weapons such as organophosphate nerve agents typically experience rapid onset of classic, easily recognizable symptoms. Despite the high mortality associated with chemical agents, practitioners usually can readily identify their effects. The effects of an exposure to biological agents, however, present later than the effects of chemical agents and often mimic other etiologies of illness. Thus, biological warfare patients present a challenge to clinicians in the recognition of victims, particularly with an aerosolized agent exposure.
In the event of a biological or a chemical bioterrorism agent attack, victims seeking care for symptoms or guidance after a possible exposure will depend on healthcare provider assistance [16]. In this review, we consider the management of a currently asymptomatic patient exposed to an unknown aerosolized substance. We focus on the evaluation and treatment of a patient with possible aerosolized biological or chemical agent exposure with slow onset. We assume the aerosol is odorless, colorless, and initially unidentified. Based on the available literature and expert opinions, we discuss optimal strategies for the diagnosis and treatment of patients with such an exposure.
METHODSPubMed and Scopus databases were searched, and the references of the included studies were also examined to find additional sources. The initial search identified 749 articles, of which 32 were included in this review. Additional references were added at the author’s discretion. Inclusion criteria were articles referencing a colorless, odorless, and aerosolized substance with an incubation period of at least 4 hours. Exclusion criteria were substances that cannot be aerosolized; substances with immediate clinical effect; and substances that are easily identifiable upon exposure due to a distinctive color, odor, taste, or texture. Only English language articles were included. We included studies such as systematic reviews, clinical guidelines, and retrospective studies. Data from the included articles were summarized and reported by agent. This article is based on previously conducted studies and does not contain any studies with human participants or animals performed by any of the authors (Supplementary Fig. 1).
RESULTSNerve agentsNovichok agentsThe relatively newly developed “Novichok,” meaning “newcomer,” or A-series nerve agents are extremely lethal, synthetic chemical weapons. These nerve agents gained notoriety after two recent cases of suspected Novichok poisonings that illustrate the extremely toxic nature of these substances. On August 20th, 2020, Alexei Navalny became seriously ill approximately 1 hour into a flight from Siberia to Moscow. The plane landed emergently in Omsk; however, officials later airlifted Alexei to Germany where tests showed Alexei had been poisoned, presumably with a Novichok agent [17]. Similarly, Sergei and Yulia Skripal were found unconscious on a park bench in Salisbury, England on March 4th, 2018. The time between possible exposure to the nerve agent and their discovery unconscious was suspected to be from 30 minutes to 3 hours [18]. A police officer who responded to the scene also developed symptoms of nerve agent poisoning and was later admitted to the hospital. One month later, the Organization for the Prohibition of Chemical Weapons (OPCW) confirmed Novichok agent poisoning [19]. In the case of both poisonings, the victims survived because of the quick and efficient actions of those involved [17,18].
These A-series nerve agents are clear, odorless liquids at room temperature and induce a cholinergic crisis by irreversibly inhibiting acetylcholinesterase, the enzyme that degrades acetylcholine. The preceding G-series nerve agents, namely sarin (GB), can cause fatal outcomes in 1 to 10 minutes; VX causes fatalities in 4 to 18 hours [20]. The precise onset of toxicity of A-series agents is not well described; however, given that A-230 is estimated to be five to eight times more toxic than VX and A-232 is estimated to be 10 times more toxic than soman (GD), the onset of symptoms may occur within minutes of inhalation [21]. Like previously developed nerve agents, the Novichok agents cause acetylcholinesterase to undergo an aging process that renders the enzyme permanently inactive if an antidote is not administered in a timely fashion [20]. These agents bind to a serine residue on acetylcholinesterase through phosphorylation. If the nerve agent is not removed via an oxime reaction, an R-alkyl group will eventually become permanently removed and produce molecular aging. The time required for aging to occur depends on the nerve agent and ranges from 2 minutes with the G-series agent soman to greater than 40 hours with VX [22]. Unlike previous nerve agents, Novichok agents also have effects at peripheral nerve synapses which can cause peripheral neuropathy [20].
Exposure to any of the Novichok agents results in a cholinergic toxidrome produced by an excess of acetylcholine at nicotinic, muscarinic, and central nervous system (CNS) receptors [21]. Nicotinic effects can include tachycardia, hypertension, diaphoresis, fasciculations, and muscle weakness. Muscarinic effects can include bradycardia, bronchorrhea, miosis, salivation, lacrimation, nausea and/or vomiting, diarrhea, and increased urination. Last, CNS effects are often the cause of death and can include seizures, respiratory paralysis, and coma [19]. Biological markers to detect acetylcholinesterase and butyrylcholinesterase activity are useful in confirming exposure to these agents [23,24]. Treatment for these agents includes 2 to 6 mg of intravenous atropine (which is a competitive antagonist at muscarinic receptors) every 5 to 10 minutes until bradycardia and/or bronchorrhea resolves, an anticonvulsant such as diazepam, and 1 to 2 grams of intravenous pralidoxime (alternative, 250 mg of obidoxime) every 3 to 6 hours (alternative, continuous infusion lasting at least 24 hours after the last atropine dose is given) [21].
Biological agentsThere are several biological agents that have the potential to be aerosolized and used as a weapon. In this review, we will discuss the most likely candidates for use in biological weaponry as categorized by the Centers for Disease Control and Prevention (CDC) (Table 1) [25], and we will review their clinical features and management.
RicinRicin is a well-documented toxin that is derived from the castor bean plant Ricinus communis. Ricin has two polypeptide chains, denoted A and B. The toxin enters the cell and exerts its effects by inhibiting protein synthesis. The British Broadcasting Corporation (BBC) journalist Georgi Markov was famously assassinated with a ricin-tipped umbrella in 1978. Saddam Hussein stockpiled the substance; and more recently, Shannon Richardson, an American actress, attempted to send ricin-laced letters to President Barack Obama [26]. Inhalation of this agent is the most dangerous route of exposure, and powdered ricin has the potential to be aerosolized. The powdered form is water soluble, odorless, and tasteless. Onset of symptoms after inhalation of ricin requires approximately 4 to 8 hours. Based on the limited data available, ricin toxicity is expected to present with allergic symptoms such as rhinorrhea and bronchospasm; flu-like symptoms including fever, nausea, cough, shortness of breath, and chest pain; as well as dyspnea and pulmonary edema. There is no readily available diagnostic test for ricin exposure, so treatment should be initiated whenever there is a suspicion of exposure. Treatment involves steroids, antihistamines, β2 adrenergic agonists for allergic symptoms, and supportive care such as continuous positive airway pressure and/or intubation for pulmonary edema [27]. Of note, there is a potential recombinant ricin toxin A subunit (RTA) vaccine, RiVax, currently undergoing US Food and Drug Administration (FDA) trials [28].
BotulismBotulism is a disease caused by toxins from Clostridium botulinum bacteria. Botulinum toxin is both the most potent toxic agent by weight known and is relatively easy to produce [29,30]. On several occasions in the 1990s, the terrorist group, Aum Shinrikyo, released aerosolized botulinum in multiple failed attacks in Tokyo [31–33]. During the same decade Iraq confessed to having generated a large arsenal of concentrated toxin [31]. Inhalation botulism incubation may be up to 72 hours.
The toxin can be aerosolized into colorless and odorless particles that are 0.1 to 0.3 μm in size [31]. After being absorbed through inhalation, ingestion, or wound penetration into the bloodstream, botulinum toxin acts at the neuromuscular junction of peripheral nerves and prevents acetylcholine release. The disease is characterized by bilateral cranial nerve dysfunction, including diplopia, ptosis, blurred vision, dysphagia, facial weakness, and nystagmus. This is accompanied by symmetrical descending weakness that progresses from the trunk to upper and lower extremities. Respiratory involvement can be caused by upper airway compromise or diaphragmatic paralysis which requires intubation and mechanical ventilation. The diagnosis is established through thorough history-taking and physical exam; antitoxin can be prophylactically administered since confirmatory testing requires 1 to 4 days for identification. Detection of the toxin is mainly made by mouse bioassay in which mice are injected with the specimen. Other techniques to detect the toxin include enzyme-linked immunosorbent assay (ELISA), mass spectroscopy, and polymerase chain reaction (PCR) testing [34,35]. The mainstay treatment of the disease is supportive therapy, respiratory care, and prompt intubation if needed. Another available therapy is heptavalent botulinum antitoxin (HBAT), which, in the United States, is requested through the CDC via local or state departments and the Strategic National Stockpile (SNS) [36]. Evidence shows reduced mortality with early administration of the antitoxin [37,38].
Anthrax
Bacillus anthracis, a gram-positive spore-forming bacteria, causes anthrax. The ability to form spores enables survival for many decades, making the spores highly resistant. In 2001, an unknown offender mailed envelopes containing anthrax spores to multiple offices causing 17 infections and four deaths in the United States [39].
Infection through spore inhalation, ingestion, or inoculation of the mucous membrane or skin results in three major anthrax syndromes: inhalation, cutaneous, and gastrointestinal anthrax [40,41]. Inhaled anthrax has the highest mortality rate of the three and is the most likely type to be used as an aerosolized biological bioterrorism agent [42]. Although usually fatal, inhalational anthrax is rare and most commonly occurs when spores are aerosolized during the processing of contaminated animal products [40]. However, inhalational anthrax is most relevant in this context because intentional aerosolization may be an act of bioterrorism [43]. The incubation period of inhaled anthrax is estimated to be 1 to 7 days [39,44]. The clinical course of infection is biphasic, with a prodromal phase lasting 4 to 5 days, beginning with nonspecific symptoms such as fever, malaise, myalgia, and progressing to more suggestive symptoms including dyspnea, hemoptysis, chest pain, and odynophagia [45]. The second phase is a rapid fulminant bacteremic phase which is manifested by the development of hypoxemia, severe respiratory distress, and shock. This may lead to death within days. Even with intensive care advancement, outcomes during the fulminant phase have not changed; however, initiating antibiotic therapy in the prodromal phase has been shown to improve outcomes [43]. Establishing a diagnosis of anthrax early is crucial because treatment initiation in a narrow, early disease window is necessary. Signs associated with inhalation anthrax include nausea, vomiting, altered mental status, cyanosis, pallor, and hematocrit >45%. Moreover, a chest x-ray demonstrating a widened mediastinum should raise clinical suspicion [46]. The CDC developed recommendations for testing patients with suspected inhalation anthrax, which include collection of blood specimens prior to antimicrobial therapy for culture and PCR testing. Also, since half of patients affected by inhalation anthrax will likely develop meningitis, lumbar puncture is performed for cerebrospinal fluid analysis. When suspicion is high, empiric treatment should include antimicrobial agents such as ciprofloxacin plus clindamycin or linezolid. If anthrax with CNS involvement is suspected, triple therapy with ciprofloxacin plus meropenem and linezolid is recommended. Penicillin G is equivalent to fluoroquinolone in anthrax strains that are susceptible. Antitoxin or anthrax immunoglobulin is an essential part of the therapeutic regimen [43]. For postexposure prophylaxis, exposed individuals are indicated to receive an anthrax vaccine and an antimicrobial drug course which includes either doxycycline or ciprofloxacin.
TularemiaTularemia, alternatively referred to as Rabbit Fever, is caused by the gram-negative zoonotic bacteria Francisella tularensis. This disease has a documented history of use in biological weapon programs and was previously stockpiled in US facilities from 1954 to 1955. These stockpiles were destroyed in 1973. Tularemia is considered as a dangerous weaponizable biologic agent due to its ability to be aerosolized and cause considerable morbidity and mortality. The clinical presentation of tularemia varies depending on the site and route of exposure, but notably causes symptoms of pneumonia after 3 to 5 days following an aerosol exposure [48]. This form of tularemia is more severe, with mortality rates of 30%. Presenting symptoms include hilar adenopathy, dry cough, shortness of breath, and chest pain. Tularemic sepsis may occur with severe infection, is often fatal, and may cause shock or disseminated intravascular congestion. Infection is confirmed via serology, and treatment includes streptomycin or gentamicin for a duration of 10 days. Alternative treatments include doxycycline, chloramphenicol, and ciprofloxacin [48].
PsittacosisWidely known as Parrot Fever, psittacosis is caused by Chlamydophila psittaci. This disease is often described in relation to bird ownership or exposure but does have the potential to be aerosolized to cause infection. The CDC has classified C. psittaci as a class B biological warfare agent for this reason. While there have been no documented uses of this agent to intentionally cause disease, the United States, the former Soviet Union, and Egypt have all researched its potential as a biological weapon. Psittacosis causes atypical pneumonia and has a nonspecific clinical presentation. The incubation period ranges from 1 to 3 days with direct local invasion of the pulmonary parenchyma, and more commonly, 7 to 15 days when a primary bacteremia occurs that leads to infection within the reticuloendothelial system. Due to the range in symptom onset, disaster medicine experts predict that a C. psittaci epidemic would result in a bimodal spike of cases, with a slightly greater second peak. Fortunately, this disease does have a lower mortality rate of roughly 20% without treatment and 1% with timely intervention. Generalized symptoms include a cough (most often a dry cough with little to no sputum), fever and malaise, vomiting and diarrhea, headache, and anorexia. Physical examination findings may include fever, rales, tachypnea, and consolidation that can be seen via chest X-ray. Serologic testing is the preferred method for diagnosis, but PCR may also be used. Oral doxycycline and tetracycline are the preferred treatments. For severely ill patients, intravenous doxycycline may be used. Azithromycin has also been shown in animal models to potentially be an effective alternative treatment (Table 2) [49].
DISCUSSIONThe availability, relatively low cost, and potential for devastating impact make chemical and biological agents increasingly appealing for those striving to inflict terror and destruction. Despite multiple attempts within the international community to limit and prevent their use, these weapons continue to appear and are more likely to be used during times of political upheaval. Given the current sociopolitical climate both nationally and abroad, clinicians need to maintain readiness for a potential attack. Particularly in the event of exposure to an unknown aerosolized substance, clinicians should be aware of the possible types of biological and chemical agent exposures and a safe approach to the asymptomatic patient. In addition, incubation periods may complicate the clinical picture as patients may not present for several days postexposure. This also underscores the importance of real-time surveillance and reporting for suspicious cases.
Before completing a thorough patient evaluation, two essential elements of patient and facility management are decontamination and containment [50]. With any aerosolized substance exposure, patients and providers must maintain airborne precautions until additional details are determined. For suspected chemical agents, providers should initiate a mass casualty decontamination protocol [51]. This may occur differently depending on the hospital facility and the agent suspected. The UK Initial Operational Response (IOR) recommends the rinse-wipe-rinse method to quickly decontaminate individuals after an exposure. An approach to the asymptomatic patient presenting to the emergency department after exposure to an unknown aerosol is shown in Fig. 1. Patients must remove all clothing to be placed in a sealed plastic bag. While many hospitals have at least small-scale showers available for decontamination, others may have larger facilities such as tents. Dry contamination using various products such as fullers earth or “blue roll” is also possible. Of note, providers should avoid dry bleach powder in cases of suspected Novichok agent exposures as it may release toxic metabolites [19].
As in any other medical evaluation, history and physical examination are fundamental, with attention to clustering of cases. Incubation periods for biological agents range from 24 hours to 16 days. Patients may not be symptomatic at the time of their evaluation or may present later without ready recall of their exposure. Diagnostic testing is minimally impactful in the assessment of nerve agent exposure. No further testing currently offers clinically relevant data pertaining to these agents specifically although new developments in mass spectrometry may change diagnostic capabilities in the future [52,53]. Potential biological markers such as phosphorylated butyrylcholinesterase are useful in detecting nerve agent exposure [24,54].
Specific microbe testing for biological agents with respiratory toxidromes is minimally available and prohibitively costly. There is a need for the development of accurate and efficient diagnostic panels or tests [55,56]. If collateral information to suggest a specific microbe exists, such as other known cases in the community, most require a specific ELISA or PCR. While some have specific toxidromes such as the neurological symptoms seen with C. botulinum or the hemorrhagic fever associated with Ebola or Marburg viruses, many of these patients initially present with symptoms of respiratory infection. Important consideration of the progression of symptoms is required as higher mortality diagnoses tend to progress more quickly.
Many factors can affect the management approach to individuals with aerosol exposure, which include the victim’s characteristics, presence of comorbidity, immune status, patterns of other known incidents, and description of the event. Moreover, the approach can differ in certain patients who are considered to have high-risk targetability such as public figures. When identified, these biological agents can be treated with antibiotics and supportive care. Most are susceptible to doxycycline and ciprofloxacin. Other agents, such as botulinum toxin, can be treated with supportive care and antitoxin whenever symptoms arise.
In the case of asymptomatic aerosol exposure, similar decontamination and containment methods should be considered alongside a high suspicion for a chemical or a biological attack [50]. Administration of atropine, pralidoxime and an anticonvulsant (diazepam) can be initiated if a cholinergic toxidrome develops during patient evaluation. In cases of massive exposure, which require large doses of atropine and pralidoxime to reverse acetylcholinesterase inhibition, a logistical plan to identify an alternative medication supply should be considered, such as deployment of the resources from the CDC SNS [57].
In the future, perpetrators may weaponize other biological agents that cannot currently be aerosolized, such as arenavirus, bunyavirus, flavivirus, marine toxin, and Orientia tsutsugamushi (scrub typhus). Similar to the development of Novichok, employing any of these agents can have catastrophic effects. Biological agents, while rarely utilized in a real-world attack, could have devastating consequences.
SUPPLEMENTARY MATERIALSupplementary material is available at https://doi.org/10.15441/ceem.22.412.
Supplementary Fig. 1.Flowchart of search results, study inclusion, and exclusion.
REFERENCES1. Wakefield B, Lewis P. Ukraine: is a chemical or biological attack likely? [Internet]. Chatham House; 2022 [cited 2022 Nov 3]. Available from: https://www.chathamhouse.org/2022/03/ukraine-chemical-or-biological-attack-likely.
2. Corera G. Russia could launch chemical attack in Ukraine: White House [Internet]. BBC News; 2022 [cited 2022 Nov 3]. Available from: https://www.bbc.com/news/uk-60683248.
3. Pazzanese C. Russia’s remaining weapons are horrific and confounding [Internet]. Harvard Gazette; 2022 [cited 2022 Nov 3]. Available from: https://news.harvard.edu/gazette/story/2022/03/harvard-analyst-assesses-chemical-weapon-threat-posed-by-russia.
4. United Nations Office for Disarmament Affairs (UNODA). Biological Weapons Convention [Internet]. UNODA; [cited 2022 Nov 3]. Available from: https://www.un.org/disarmament/biological-weapons.
5. Organisation for the Prohibition of Chemical Weapons (OPCW). Chemical Weapons Convention [Internet]. OPCW; [cited 2022 Nov 3]. Available from: https://www.opcw.org/chemical-weapons-convention.
6. Lyon RF. The COVID-19 response has uncovered and increased our vulnerability to biological warfare. Mil Med 2021; 186:193-6.
7. United Nations Counter-Terrorism Centre (UNCCT). Chemical biological, radiological and nuclear terrorism [Internet]. UNCCT; [cited 2022 Nov 3]. Available from: https://www.un.org/counterterrorism/cct/chemical-biological-radiological-and-nuclear-terrorism.
9. Barras V, Greub G. History of biological warfare and bioterrorism. Clin Microbiol Infect 2014; 20:497-502.
10. Guillemin J. Crossing the normative barrier: Japan’s biological warfare in China in World War II; In: Lentzos F, editor. Biological threats in the 21st century. Imperial College Press; 2016. p.17-40.
11. Haines DD, Fox SC. Acute and long-term impact of chemical weapons: lessons from the Iran-Iraq War. Forensic Sci Rev 2014; 26:97-114.
13. Tu AT. The use of VX as a terrorist agent: action by Aum Shinrikyo of Japan and the death of Kim Jong-Nam in Malaysia: four case studies. Glob Secur Health Sci Policy 2020; 5:48-56.
14. Okumura T, Hisaoka T, Yamada A, et al. The Tokyo subway sarin attack: lessons learned. Toxicol Appl Pharmacol 2005; 207(2 Suppl):471-6.
15. Cronin AK. Terrorist motivations for chemical and biological weapons use: placing the threat in context. Def Secur Anal 2004; 20:313-20.
16. Sharififar S, Jahangiri K, Zareiyan A, Khoshvaghti A. Factors affecting hospital response in biological disasters: a qualitative study. Med J Islam Repub Iran 2020; 34:21.
17. BBC Russian. Alexei Navalny: two hours that saved Russian opposition leader’s life [Internet]. BBC News; 2020 [cited 2022 Nov 3]. Available from: https://www.bbc.com/news/world-europe-54012278.
18. BBC Russian. Russian spy: what happened to Sergei and Yulia Skripal? [Internet]. BBC News; 2018 [cited 2022 Nov 3]. Available from: https://www.bbc.com/news/uk-43643025.
19. Chai PR, Hayes BD, Erickson TB, Boyer EW. Novichok agents: a historical, current, and toxicological perspective. Toxicol Commun 2018; 2:45-8.
20. Agency for Toxic Substances and Disease Registry (ATSDR). ToxFAQs for nerve agents (GA, GB, GD, VX) [Internet]. ATSDR; 2014 [cited 2022 Nov 3]. Available from: https://wwwn.cdc.gov/TSP/ToxFAQs/ToxFAQsDetails.aspx?faqid=524&toxid=93.
21. Franca TC, Kitagawa DA, Cavalcante SF, da Silva JA, Nepovimova E, Kuca K. Novichoks: the dangerous fourth generation of chemical weapons. Int J Mol Sci 2019; 20:1222.
22. Hulse EJ, Haslam JD, Emmett SR, Woolley T. Organophosphorus nerve agent poisoning: managing the poisoned patient. Br J Anaesth 2019; 123:457-63.
23. Wille T, Djordjevic S, Worek F, Thiermann H, Vucinic S. Early diagnosis of nerve agent exposure with a mobile test kit and implications for medical countermeasures: a trigger to react. BMJ Mil Health 2020; 166:99-102.
24. Black RM, Read RW. Biological markers of exposure to organophosphorus nerve agents. Arch Toxicol 2013; 87:421-37.
25. Centers for Disease Control and Prevention (CDC). Emergency preparedness and response: bioterrorism agents/diseases [Internet]. CDC; 2018 [cited 2022 Nov 10]. Available from: https://emergency.cdc.gov/agent/agentlist-category.asp.
26. BBC News. Actress Shannon Richardson pleads guilty in ricin case [Internet]. BBC News; 2013 [cited 2022 Nov 3]. Available from: https://www.bbc.com/news/world-us-canada-25326964.
27. Yun BJ. Ricin toxin from Ricinus communis (castor bean) attack; In: Ciottone GR, editor. Ciottone’s disaster medicine. 2nd ed. Elsevier; 2016. p.804-6.
28. Toth RT 4th, Angalakurthi SK, Van Slyke G, et al. High-definition mapping of four spatially distinct neutralizing epitope clusters on RiVax, a candidate ricin toxin subunit vaccine. Clin Vaccine Immunol 2017; 24:e00237-17.
29. Johns Hopkins Bloomberg School of Public Health. Johns Hopkins Working Group on Civilian Biodefense says botulinum toxin is a major biological weapons threat [Internet]. Johns Hopkins Bloomberg School of Public Health; 2001 [cited 2022 Nov 6]. Available from: https://publichealth.jhu.edu/2001/botulinum-toxin-release-2001.
30. Karwa M, Currie B, Kvetan V. Bioterrorism: preparing for the impossible or the improbable. Crit Care Med 2005; 33(1 Suppl):S75-95.
31. Arnon SS, Schechter R, Inglesby TV, et al. Botulinum toxin as a biological weapon: medical and public health management. JAMA 2001; 285:1059-70.
33. Sugishima M. Aum Shinrikyo and the Japanese law on bioterrorism. Prehosp Disaster Med 2003; 18:179-83.
34. Hodowanec A, Bleck TP. Botulism (Clostridium botulinum); In: Bennett JE, Dolin R, Blaser MJ, editors. Mandell, Douglas, and Bennett’s principles and practice of infectious diseases. 8th ed. Vol. 2; Elsevier; 2015. p.2763-7.
35. Solomon HM, Lilly T Jr. BAM chapter 17: Clostridium botulinum [Internet]. FDA; 2001 [cited 2022 Nov 6]. Available from: https://www.fda.gov/food/laboratory-methods-food/bam-chapter-17-clostridium-botulinum.
36. Centers for Disease Control and Prevention (CDC). Investigational heptavalent botulinum antitoxin (HBAT) to replace licensed botulinum antitoxin AB and investigational botulinum antitoxin E. MMWR Morb Mortal Wkly Rep 2010; 59:299.
37. Yu PA, Lin NH, Mahon BE, et al. Safety and improved clinical outcomes in patients treated with new equine-derived heptavalent botulinum antitoxin. Clin Infect Dis 2017; 66(suppl_1):S57-64.
38. O’Horo JC, Harper EP, El Rafei A, et al. Efficacy of antitoxin therapy in treating patients with foodborne botulism: a systematic review and meta-analysis of cases, 1923-2016. Clin Infect Dis 2017; 66(suppl_1):S43-56.
39. Jernigan DB, Raghunathan PL, Bell BP, et al. Investigation of bioterrorism-related anthrax, United States, 2001: epidemiologic findings. Emerg Infect Dis 2002; 8:1019-28.
41. Brachman PS, Kaufmann AF. Anthrax; In: Brachman PS, Abrutyn E, editors. Bacterial infections of humans: epidemiology and control. Springer; 2009. p.105-19.
42. Inglesby TV, Henderson DA, Bartlett JG, et al. Anthrax as a biological weapon: medical and public health management. Working Group on Civilian Biodefense. JAMA 1999; 281:1735-45.
43. Centers for Disease Control and Prevention (CDC). Update: Investigation of bioterrorism-related anthrax and interim guidelines for clinical evaluation of persons with possible anthrax. MMWR Morb Mortal Wkly Rep 2001; 50:941-8.
44. Meselson M, Guillemin J, Hugh-Jones M, et al. The Sverdlovsk anthrax outbreak of 1979. Science 1994; 266:1202-8.
45. Holty JE, Bravata DM, Liu H, Olshen RA, McDonald KM, Owens DK. Systematic review: a century of inhalational anthrax cases from 1900 to 2005. Ann Intern Med 2006; 144:270-80.
46. Kyriacou DN, Stein AC, Yarnold PR, et al. Clinical predictors of bioterrorism-related inhalational anthrax. Lancet 2004; 364:449-52.
47. Evans ME, Gregory DW, Schaffner W, McGee ZA. Tularemia: a 30-year experience with 88 cases. Medicine (Baltimore) 1985; 64:251-69.
48. Francisella tularensis (Tularemia) attack [Internet]. ClinicalKey; [cited 2022 Nov 10]. Available from: https://www-clinicalkey-com/content/book/3-s2.0-B9780323286657001266.
49. Chlamydophila psittaci (Psittacosis) attack [Internet]. ClinicalKey; cited [2022 Nov 10]. Available from: https://www-clinicalkey-com.proxygw.wrlc.org/#!/content/book/3-s2.0-B9780323286657001382
.
50. Kumar V, Goel R, Chawla R, Silambarasan M, Sharma RK. Chemical, biological, radiological, and nuclear decontamination: recent trends and future perspective. J Pharm Bioallied Sci 2010; 2:220-38.
51. Collins S, James T, Carter H, et al. Mass casualty decontamination for chemical incidents: research outcomes and future priorities. Int J Environ Res Public Health 2021; 18:3079.
52. Moshiri M, Darchini-Maragheh E, Balali-Mood M. Advances in toxicology and medical treatment of chemical warfare nerve agents. Daru 2012; 20:81.
53. John H, Thiermann H. Poisoning by organophosphorus nerve agents and pesticides: an overview of the principle strategies and current progress of mass spectrometry-based procedures for verification. J Mass Spectrom Adv Clin Lab 2021; 19:20-31.
54. van der Schans MJ, Polhuijs M, van Dijk C, et al. Retrospective detection of exposure to nerve agents: analysis of phosphofluoridates originating from fluoride-induced reactivation of phosphylated BuChE. Arch Toxicol 2004; 78:508-24.
55. Hosseini-Shokouh SJ, Sheikhi RA, Hosseini SM, Moradimajd P. The biological weapons threats and coping strategies for health promotion. J Educ Health Promot 2021; 10:127.
Fig. 1.Approach to asymptomatic patient presentation in the emergency department with exposure to unknown aerosol substance. CDC, Centers for Disease Control and Prevention; PPE, personal protective equipment; IV, intravenous; 2-PAM, pralidoxime. 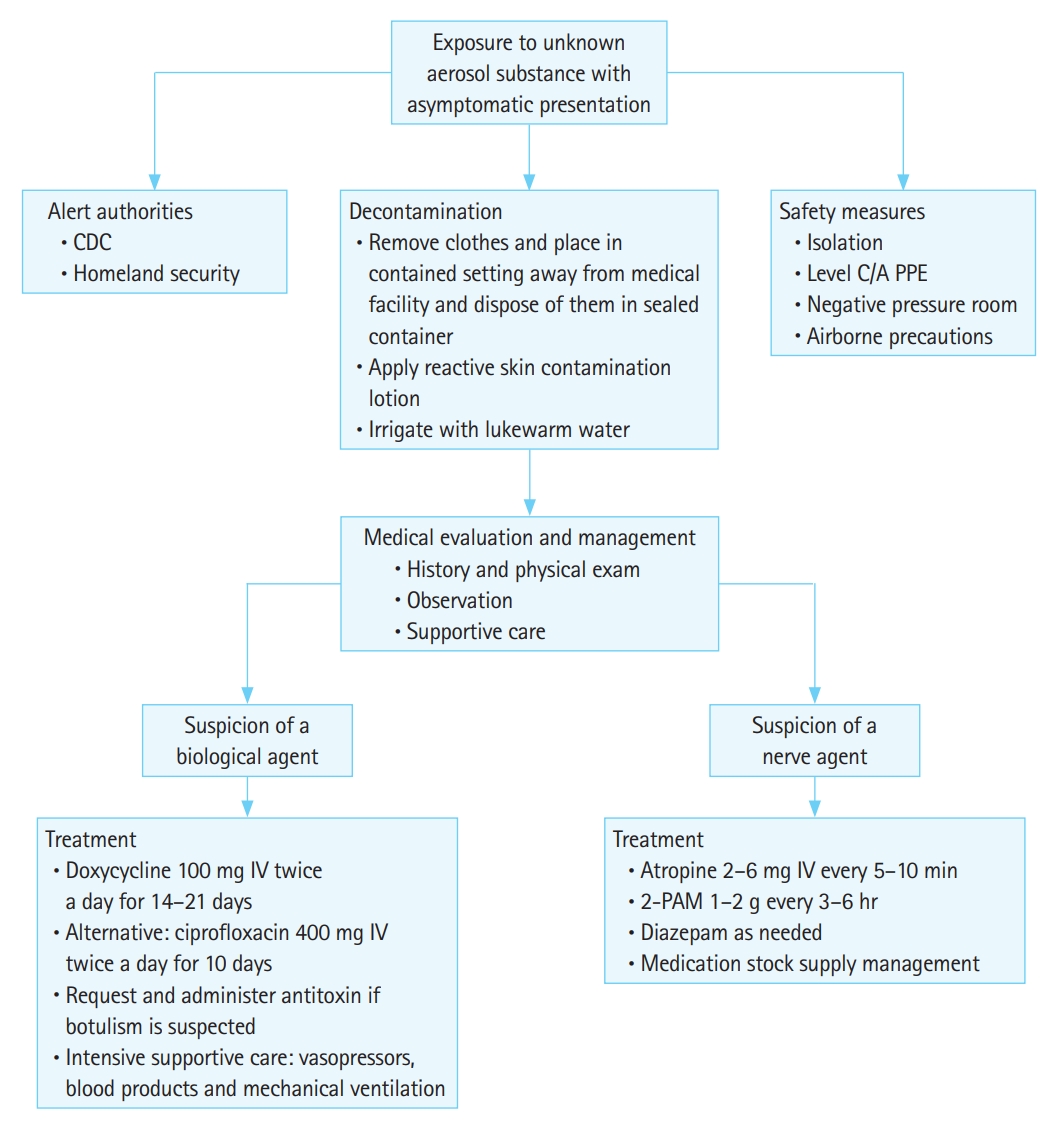
Table 1.Bioterrorism agents categorized by CDC
Table 2.Summary of agent mechanism of action, clinical presentation, and treatment SLUDGEM, salivation, lacrimation, urination, defecation, gastric hypermotility, emesis, and miosis; IV, intravenous; CPAP, continuous positive airway pressure; HBAT, heptavalent botulinum antitoxin; cAMP, cyclic adenosine monophosphate; MAP, mitogen activated protein; CNS, central nervous system; DIC, disseminated intravascular coagulation. |
|
||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||